Macro ATR-FT-IR Spectroscopic Imaging of Dynamic Processes
Spectroscopy
Macro attenuated total reflection FT-IR spectroscopic imaging is a powerful and underutilized tool. This article presents an overview of approaches and opportunities for using this method to study dynamic processes such as diffusion, sorption, crystallization, and dissolution.
Macro attenuated total reflection (ATR)-Fourier transform infrared (FT-IR) spectroscopic imaging is a powerful analytical tool, but it is still underexploited in the spectroscopic imaging community. This article presents an overview of current macro ATR-FT-IR imaging approaches and opportunities for studying dynamic processes such as diffusion, sorption, crystallization, and dissolution.
Fourier transform infrared (FT-IR) spectroscopic imaging combines the chemical specificity of IR spectroscopy with the spatially resolved nature of a mercury cadmium telluride (MCT) focal plane array (FPA) detector. These detectors typically contain 4096 (64 × 64) or 16,384 (128 × 128) infrared sensitive elements or pixels. Unlike using single-element detectors, where the resulting spectrum is averaging information from different parts of the sample, each detector element in an FPA is focused on a different spatial region of the sample. For heterogeneous samples, this increases sensitivity because with a single-element detector, a small particle may be "averaged out" by the presence of other materials in a single spectrum, but with imaging, such a particle would occupy a few pixels in the array, whose spectra would then be that of only the particle and not the surrounding material (1). The spatially resolved nature of imaging also allows for location-based spectral processing from simple masking to improve average spectra, to clustering or classification techniques to identify regions of interest (2). It is important to note that the use of the term imaging indicates that spectra are obtained from all pixels simultaneously in contrast to mapping, where a single-element detector is scanned over the desired area. The main advantage of imaging compared to mapping is the ability to study dynamic systems and samples as spectra can be obtained within a few minutes or less (3). Advances in computational power and optimization of scanning parameters have enabled images to be obtained within 50 ms, fast enough to study moving droplets within a microfluidic device (4).
Imaging is commonly achieved in transmission, in which the IR beam passes through a sample that is on one or between two IR-transparent windows. The principal disadvantages of transmission are that the sample must be thin (<20 μm) and aqueous samples are particularly challenging to study because of the strong absorption of water. Attenuated total reflection (ATR) mode uses a suitably shaped crystal of high refractive index material, in which incident infrared light undergoes total internal reflection at the interface between the crystal and the sample. At this interface, an evanescent wave penetrates into the sample, whose depth of penetration (dp) depends on the angle of incidence (θ), refractive indexes of the crystal (n1) and sample (n2), and wavelength (λ) of infrared light. The depth of penetration, where the intensity of the electric field of incident infrared light falls below e-1 of the maximum value is given below:
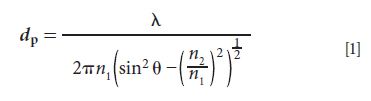
Figure 1: Schematic of macro ATR-FT-IR imaging showing an (a) FT-IR spectrometer with redirection of the infrared beam to the macrochamber, (b) through an ATR accessory, and on to the FPA detector. The FPA records all spectra simultaneously, producing (c) a spectral data-cube where peak integration on bands corresponding to unique components, can be applied to all spectra. Fast scanning allows many spectral data cubes to be obtained over time, and (d) plotting integrated absorbance values against a color scale for all pixels and for all chosen integration bands from each data-cube results in a series of images, showing the spatial distributions of these components.
For mid-IR, the depth of penetration is typically 1–5 μm and the crystal material used depends on the type of sample and material properties required of the crystal. Germanium and zinc selenide can be damaged easily with hard samples, with the latter also having a limited pH range for samples. The refractive indexes of these materials are 4 and 2.4, respectively, with germanium commonly used in microscope ATR objectives because of its high numerical aperture. Silicon and diamond are also commonly used materials, with diamond being particularly robust. They have refractive indexes of 3.4 and 2.4, respectively.
The FPA detector is often mounted in a separate compartment or macrochamber attached to an FT-IR spectrometer in which different ATR accessories can be positioned. The type of accessory can vary depending on the field of view required and the angle of incidence of the incoming beam (which affects depth of penetration). The most suitable accessory depends on the experiment and different types are demonstrated in the following sections. The FT-IR spectrometer sends the IR beam through one of its side-ports, through the macrochamber and ATR accessory, to the FPA where the IR beam is focused on the surface of the crystal and FPA to obtain spatially resolved IR spectra. It should be noted that because of the exponential decay of the evanescent field, particles can appear smaller than their true size (5), but for many applications, the interest is in chemical and spatial changes at the surface. To obtain two-dimensional (2D) color images, univariate analysis, which involves selecting spectral bands unique to components of interest, can be performed on the spectral data cube. Plotting the integrated absorbance of spectral bands of different components for all pixels against a color scale produces images with respect to each component, where regions of differing concentration can be identified by different colors. A summary of this process is shown in Figure 1. Multivariate approaches are often used when many bands overlap such as in complex pharmaceutical systems (6,7). This versatile approach can also be applied in micro mode with an IR microscope (8), though the focus of this article is on macro-mode imaging (9). Macro-mode imaging has many applications (9,10), and this article provides examples of macro-ATR accessories and case studies where their different properties were used to reveal new information about the samples under investigation.
Overview of ATR Spectroscopic Accessories
Figure 2: Schematic of a single-reflection large field of view macro-ATR accessory that is being used to study protein crystallization (11) in this example. The chemical image below the schematic is of protein (thaumatin) crystals formed on the measuring surface of the ZnSe accessory; the size of the image is 2.56 mm. The chemical image is adapted and reprinted with permission from reference 11. Copyright 2009 American Chemical Society.

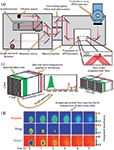
Large field of view accessories use no magnifying optics and, thus, have a field of view between 2.5 mm × 3.6 mm (11) and 15.4 mm × 21.4 mm2 (12), depending on the pixel size and number of pixels in the FPA (64 × 64 up to 128 × 128), the angle of incidence, and the accessory used. The Specac Oil Analyser (13), FastIR (14), and Pike Veemax 2 (15) accessories (the latter of which has an adjustable angle of incidence) are examples of large field of view accessories. Schematics of different accessories are shown in Figures 2 and 3. In some accessories, an undesirable gradient of absorbance can be observed along the crystal as the optical configuration permits a spread of different angles of incidence, affecting the depth of penetration (14). In other accessories with movable mirrors, this gradient can be minimized or removed completely (10). Crystals used with such accessories are often zinc selenide, silicon, or germanium. Large field of view accessories are used when spatial resolution is secondary to field of view and, as such, are suitable for pharmaceutical studies (16), microfluidics (17), fingerprint analysis (18), depth profiling (15), protein crystallization (19), and other high-throughput approaches. Because the crystals are not as robust as diamond, they cannot be used for high-pressure experiments or in cases where materials need to be compacted against the crystal surface for good contact. The use of acidic or strong bases can also damage zinc selenide, leaving silicon as the only suitable option. However, silicon's higher refractive index than zinc selenide reduces the depth of penetration, resulting in a weaker probing depth, and its higher cut-off precludes the study of spectral bands below ~1000 cm-1. The use of depth profiling by changing the angle of incidence is particularly useful for chemically imaging thin polymer films (15) and has potential in the future for depth profiling other materials, overcoming one of the limitations of transmission. The Oil Analyser has a field of view of 2.5 mm × 3.6 mm with a 64 × 64 pixel detector (11). The Pike VeeMax has a field of view of 7.0 mm × 9.8 mm with a silicon crystal (19) or 5.3 mm × 7.9 mm with ZnSe crystal (20), depending on the angle of incidence, when using a 96 × 96 pixel detector. An expanded field of view accessory as shown in Figure 4 is particularly useful for high-throughput studies in which a larger area (15.4 mm × 21.4 mm) of the ATR crystal can be imaged (12). For an angle of incidence of 45°, images will have an aspect ratio of 1:√2 unless correcting optics are used, and other angles will produce images of different aspect ratios as shown in Figure 3.
Figure 3: Schematic of a variable-angle large field of view macro-ATR accessory that is being used to image a thin film of PVP surrounded by PDMS in this example (15). The chemical images below show PVP on the left side, and PDMS on the right side of each image. The effect of increasing the angle of incidence changes the aspect ratio and the integrated absorbance of PDMS decreases, especially on the left side as the evanescent wave only interacts with PVP and not PDMS at higher angles of incidence.
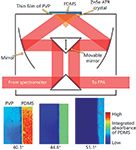
The imaging Golden Gate accessory, initially developed by Specac for nonimaging applications and, thereafter, containing aspect-ratio correcting optics for imaging, contains a diamond ATR crystal mounted in tungsten carbide (Figure 5). The diamond is cut to form a 45° inverted prism with a top surface of 2 mm × 2 mm. Lenses, normally zinc selenide, condense the incident beam into the diamond, and a similar lens on the opposite side expands the beam, resulting in an imaging area of approximately 656 μm × 579 μm for a 64 × 64 FPA detector, a magnification of approximately 4×. It is important to note that with noncorrecting optics, the imaging area is 820 μm × 1140 μm (21). This accessory allows for in situ compaction and dissolution of pharmaceutical tablets (22), high-pressure and high-temperature experiments (23), and, more recently, the use of apertures for variable angles of incidence to eliminate dispersion artefacts because of high refractive index samples (24). The use of apertures in this system allows for depth profiling in a similar manner to that of the large field of view accessories (25).
Dynamic Applications with Macro ATR-FT-IR Spectroscopic Imaging
Pharmaceutical Tablet Dissolution
Figure 4: Photograph of an expanded field of view macro-ATR accessory using a red laser beam for demonstration.

Drug release from solid oral dosage forms is one of the most common methods by which drugs are administered to patients, and the release mechanisms that occur after a tablet is ingested determine the bioavailability of the drug. Drugs or active pharmaceutical ingredients (APIs) are often complex molecules with low aqueous solubilities, so one important role of other components or excipients within the tablet is to aid solubilization of the drug as it is released from the tablet. Conventional dissolution studies use United States Pharmacopeia (USP) dissolution tests, in which tablets are dissolved under well-defined flow conditions and the amount of dissolved drug is measured using ultraviolet–visible (UV–vis) spectroscopy. This approach has drawbacks such as not giving information about mechanisms within the tablet as the drug is released, and it only shows the amount of drug dissolved over time, not necessarily released in other solid forms such as particles or precipitates. This distinction is important as the drug can be released from the tablet, but in a form which is not bioavailable (that is, a solid or undissolved form). Other approaches such as UV imaging (26), Raman (27), and magnetic resonance imaging (MRI) (28,29) can also be used noninvasively to obtain information from a tablet as it dissolves. MRI can give three-dimensional (3D) images of the whole tablet under USP IV conditions, but its acquisition times for each 3D image are long, up to 30 min (28). FT-IR spectroscopic imaging of tablets can be done in transmission mode, but the tablets must be very thin to reduce the effects of strong water absorbance (although the use of D2O can aid with this) (30) and formulated to reduce scattering of the infrared light, limiting the applicability of this approach in studying pharmaceutically relevant tablets.
Figure 5: Schematic of a diamond ATR accessory being used to analyze a pharmaceutical tablet dissolving in a flow cell in this example (32).
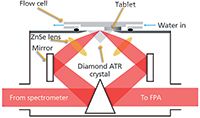
Macro ATR-FT-IR spectroscopic imaging is particularly suited to studying drug release from microparticles (31) and tablets (32) because they do not need to be thin and components within the layer in contact with the crystal can be imaged over time (33). Importantly, spectroscopic imaging can reveal drug crystallization because of changes in molecular interactions (16). Using a diamond ATR accessory, the tablet can be compacted in situ, guaranteeing good contact with the crystal and with a suitable flow cell (34), dissolution can also be carried out without removing the tablet from the crystal. A UV–vis detector connected in-line downstream can give the amount of dissolved drug over time in a comparable way to USP and using a transparent flow cell (with ex situ compaction), visible images can also be captured (32). With the latter approach, differences have been demonstrated between visibly observed dissolution fronts (that is, water penetration into the tablet, drug dissolution and diffusion, and so on) and those produced from FT-IR imaging. This result was particularly important because relying on changes in refractive index or color changes is not a reliable method to study dissolution and it would only be applicable to certain formulation types. It is also possible to use a large field of view ZnSe variable angle accessory for an overall view of the tablet as it dissolves. This approach has been combined with computer modeling of drug release, where information from FT-IR imaging was used for model validation and predictive capability testing (35). The combination of computer modeling and FT-IR imaging opens up the possibility of formulation optimization for tablets of different shapes and composition (20).
Protein Crystallization and Adsorption
The function of a protein is governed by its tertiary or 3D structure, and X-ray crystallography is usually used to obtain structural information. However, crystallization is a major bottleneck in protein screening because the conditions under which crystals will form and grow are narrow. Crystallization normally occurs as the solution is brought to super-saturation and nucleation followed by crystal growth. However, excessive nucleation can cause crystals to be too small and have insufficient space to grow into long-range ordered structures, or the protein may precipitate into an amorphous solid. Thus, it is necessary to optimize the crysallization conditions based on temperature, pH, and the presence of other chemicals or "leads" whose function is to initiate crystallization or stabilize growing crystals such that they become large enough for X-ray crystallography. One problem is that other species crystallize in addition to the protein, and determining if a particular crystal is proteinaceous is often chemically or physically destructive.
Consequently, it is desirable to have a chemically specific, nondestructive method that can determine if a crystal is a protein and if possible, obtain structural information at the same time. UV fluorescence imaging can be used, but with wide absorption bands, it is not as chemically specific as IR spectroscopy. In addition, IR spectroscopy allows for the determination of secondary structure. The crystallization of proteins directly onto an ATR crystal, combined with imaging, allows for determining which crystals are indeed proteins and how quickly they grow over time. Unlike Raman spectroscopy, where individual crystals would have to be located and scanned, macro ATR-FT-IR imaging obtains spectra from all protein crystals simultaneously. The crystallization of lysozyme has been demonstrated on a ZnSe ATR crystal, and the use of this large field of view also opens up the possibility for high-throughput approaches, for example, different concentrations of NaCl and protein solution can be imaged over time in microwells (11). However, the spatial resolution suffers with a large field of view, and smaller protein crystals may not be detected. The use of a diamond ATR crystal (11) demonstrated protein crystallization at a higher spatial resolution, and combining this crystal with a microfluidic approach (36) would enable high-throughput screening of different conditions, considerably speeding up the discovery of the optimum set of parameters.
Another example of a high-throughput approach is the surface modification of the ATR crystal itself (19). In this example, a gradient of hydrophobicity was created using a modified vapor deposition approach, in which octyltrichlorosilane is deposited on a silicon ATR crystal in one direction. It was shown that lysozyme adsorption and crystallization decreased as the hydrophobicity decreased along the crystal. The use of an FPA detector allowed for pixel binning in the direction normal to the gradient on the crystal surface, significantly improving the signal-to-noise ratio. Surface modification with nanoparticles for surface-enhanced infrared absorption spectroscopy (SEIRAS) can increase sensitivity by two orders of magnitude and enables the study of monolayers (37). Nanoparticle layer thickness and uneven deposition can present a problem with loss of spectral intensity and uneven enhancement. However, there exists great potential for imaging in this area and though this has been applied to polymers (10), it has yet to be applied to proteins.
Supercritical CO2 Interaction with Polymers
Another exciting application of macro ATR-FT-IR imaging is for the analysis of materials processed with supercritical fluids. Supercritical CO2 is a widely used supercritical solvent because it is nontoxic, environmentally benign, and relatively inexpensive. It can also be removed from systems at ambient temperature and pressure unlike many organic solvents because it is a gas under these conditions. An important effect is the plasticization of polymers in which the supercritical CO2 is absorbed into the polymer, causing it to swell and the glass transition temperature (Tg) to be lowered. This aids the diffusion of materials into the polymer and the processing of the polymer itself via extrusion or blending (38,39). Using macro ATR-FT-IR imaging with a high-pressure cell presents a unique opportunity to study polymer behavior under high pressures of CO2. In an article demonstrating this approach (40), two polymer systems were studied. The first was a polystyrene–polyvinylmethylether blend, which has been observed to have a lower critical solution temperature, thus increases in temperature result in phase separation. In this system, phase separation was observed after pressurizing the polymer blend with CO2 to 60 bar and back to atmospheric; the CO2 plasticized the polymers allowing phase separation to occur. The imaging approach allowed in situ observation of the formation of polystyrene- or polyvinylmethylether-rich regions, demonstrating the effect of supercritical CO2 in a similar way to raising the temperature. The second was a study into the sorption of supercritical CO2 into polymethylmethacrylate (PMMA) and polyethylene oxide (PEO) films simultaneously at different temperatures. It was observed that CO2 solubility decreases as a function of increasing temperature with PMMA. For PEO, little sorption was observed at 30 °C, but at 50 °C after the polymer had melted, the absorbed CO2 concentration increased significantly. This demonstrated a high-throughput approach for analyzing different polymers under identical environmental conditions and with imaging, it was also possible to observe polymer interdiffusion.
Asphaltene Formation and Deposition
Heating of crude oil for distillation accounts for a large fraction of energy use during oil refining, and fouling caused by precipitating species is a major source of inefficiency (41). Fouling materials are complex, containing insoluble impurities, inorganic species, and asphaltenes, which are polycyclic aromatic hydrocarbons, one of the heaviest fractions of crude oil. Asphaltenes are a major contribution to fouling and their composition and tendency to form depend on the type of crude oil, temperature, and pressure of the system. There are many analytical approaches for studying asphaltenes, each with their own advantages and drawbacks. FT-IR single-element spectroscopy has been used to study petroleum asphaltenes (42); however, with spectroscopic imaging it is possible to analyze crude oil behavior under different or industrially relevant conditions to observe asphaltene formation and obtain chemical information from their deposits in situ. Using a diamond ATR accessory, solid crude oil deposits were initially studied and the use of custom apertures in this accessory removed anomalous dispersion, enabling different chemical clusters to be observed within the deposit (24). Applying this method to study deposition in situ with crude oil was achieved by Gabrienko and colleagues (43), where n-heptane was used to precipitate out asphaltenes. The approach demonstrated the viability of using in-situ macro ATR-FT-IR imaging to study these deposits as they formed over time, and it was subsequently used to study asphaltene deposition from different geographical sources of crude oil (44). From this, it appeared that the total asphaltene concentration increased with increasing sulfur content (45) with sulfoxides observed as the dominant sulfur species, but other sulfur species that may be present have spectral bands beyond the lower limit of the FPA detector (<900 cm-1) (46). Further investigations into the stability of oils under different conditions such as temperature and pressure are possible with an imaging diamond ATR accessory (43,44), attesting to the versatility of this approach to studying these samples. Thus, in the future, the information gained can be used to find optimal physical or chemical conditions to prevent asphaltene deposition.
Microfluidics
Microfluidic technology continues to expand as an analytical tool because of the ability to manipulate very small volumes (up to microliters) of fluids under conditions where laminar flow and diffusive heat and mass transfer tend to dominate. The importance of label-free methodologies such as FT-IR imaging as a detection tool has recently been discussed by us in this publication (47). This miniaturization and control of flow allow for so-called "lab on a chip" devices to be manufactured. A popular material for manufacturing microfluidic devices is polydimethylsiloxane (PDMS), in which channels can be created using a variety of processes such as soft lithography (48). Direct printing using paraffin waxes has also been proven to be particularly effective for rapid prototyping of devices. With this method, it is possible to print microfluidic channels directly onto infrared transparent windows (49), enabling analysis of reactions by FT-IR spectroscopic imaging (50). However, there is the problem of high water absorption in these transmission devices. Printing wax directly onto an ATR crystal (49), or using a suitable PDMS device, removes this caveat and has allowed mixing of fluids such as PEG and water, or H2O and D2O to be observed (17). In these instances, a "Y"-shaped device was used with two fluids entering at the top of the "Y" and mixing, establishing steady-state conditions along the channels. Imaging allows the progression of chemical mixing (in the case of PEG and water) and reaction (in the case of H2O and D2O) along the channel to be monitored, with a gradient being established in the former case, or the reaction products (HDO) in the latter case formed at the interface between the two input fluids. Under steady-state conditions, the ATR crystal can be moved so as to map a larger area of the microfluidic device (17). Fast imaging in transmission has been achieved using "kinetic mode," where interferograms are obtained and saved without coaddition, enabling spectral images to be acquired in as little as 50 ms (4). The speed of acquisition is affected by spectral resolution (which determines the distance the moving mirror has to move in the interferometer), the spectral range, and the refresh rate of the FPA. The latter depends on the FPA window size, hence smaller window sizes (64 × 64) will scan faster than larger sizes (128 × 128). This fast scanning is important for imaging segmented flows, in which one phase is separated by another. This allows each droplet to become a "micro-batch reactor" and eliminates axial flow within the channel. In ATR mode, the droplet phase can become detached from the ATR crystal surface, although this can be mitigated by reducing the flow velocity. Monitoring these droplets as they progress through the device allows the reaction progress to be analyzed, and changing the input chemistry of each droplet as it is generated facilitates a high-throughput approach for measuring and optimizing reaction chemistry (47,50).
Conclusions
In this article, several case studies were presented that demonstrate the versatility of macro ATR-FT-IR spectroscopic imaging. The selection of the appropriate accessory, crystal, and angle of incidence depends on the system being studied and also on the required spatial resolution, field of view, sample type, and desired experimental conditions. As ATR-FT-IR spectroscopy relies on the evanescent field, it does not suffer from strong absorption of water, or other strongly absorbing samples, which can obscure weaker bands and enables a variety of aqueous or other systems to be quantitatively studied. The FPA detector increases sensitivity for heterogeneous samples as each pixel corresponds to a unique location on the sample surface. This feature is particularly powerful when combined with a macro-ATR approach for analyzing pharmaceutical drug or protein crystallization, asphaltene deposition, phase separation, and fluid analysis in microfluidic devices. This approach also facilitates high-throughput analysis, where many different samples can be analyzed under identical conditions, increasing experimental reliability and reducing parameter space exploration times. The time-resolved nature enables dynamic systems to be imaged in-situ, where depending on the spectral quality and range required, spectral data cubes can be obtained within 50 ms. As macro ATR-FT-IR imaging can analyze a variety of dynamic samples and systems with high resolution and chemical specificity, its applications in emerging areas such as biotechnology, supercritical fluids, microfluidics, and novel formulation and material design are far-reaching.
Acknowledgments
S.G. Kazarian acknowledges research funding from the European Research Council under the European Community's Seventh Framework Programme (FP7/2007-2013)/ERC advanced grant agreement no. (227950).
References
(1) K.L.A. Chan and S.G. Kazarian, Analyst 131, 126–131 (2006).
(2) P. Lasch, W. Haensch, D. Naumann, and M. Diem, Biochim. Biophys. Acta. 1688, 176–186 (2004).
(3) K.M. Dorling and M.J. Baker, Trends Biotechnol. 31, 437–438 (2013).
(4) K.L.A. Chan, X. Niu, A.J. deMello, and S.G. Kazarian, Anal. Chem. 83, 3606–3609 (2011).
(5) N.J. Everall, I.M. Priestnall, F. Clarke, L. Jayes, G. Poulter, D. Coombs, and M.W. George, Appl. Spectrosc. 63, 313–320 (2009).
(6) J. Tetteh, K.T. Mader, J.M. Andanson, W.J. McAuley, M.E. Lane, J. Hadgraft, S.G. Kazarian, and J.C. Mitchell, Anal. Chim Acta. 642, 246–256 (2009).
(7) J. van der Weerd and S.G. Kazarian, J. Pharm. Sci. 94, 2096–2109 (2005).
(8) H.M. Pollock and S.G. Kazarian, in Encyclopedia of Analytical Chemistry, (John Wiley & Sons, Ltd, 2014), pp. 1–26, DOI: 10.1002/9780470027318.a5609.pub2.
(9) S.G. Kazarian and K.L.A. Chan, Appl. Spectrosc. 64, 135A–152A (2010).
(10) S.G. Kazarian and K.L.A. Chan, Analyst 138, 1940–1951 (2013).
(11) K.L.A. Chan, L. Govada, R.M. Bill, N.E. Chayen, and S.G. Kazarian, Anal. Chem. 81, 3769–3775 (2009).
(12) S.G. Kazarian, Anal. Bioanal. Chem. 388, 529–532 (2007).
(13) J. van der Weerd and S.G. Kazarian, Appl. Spectrosc. 58, 1413–1419 (2004).
(14) E. Wessel, G. Heinsohn, H. Schmidt-Lewerkuehne, K.-P. Wittern, C. Rapp, and H.W. Siesler, Appl. Spectrosc. 60, 1488–1492 (2006).
(15) K.L.A. Chan and S.G. Kazarian, Appl. Spectrosc. 61, 48–54 (2007).
(16) S.G. Kazarian and K.L.A. Chan, Macromolecules 36, 9866–9872 (2003).
(17) K.L.A. Chan, S. Gulati, J.B. Edel, A.J. de Mello, and S.G. Kazarian, Lab Chip 9, 2909–2913 (2009).
(18) C. Ricci, P. Phiriyavityopas, N. Curum, K.L.A. Chan, S. Jickells, and S.G. Kazarian, Appl. Spectrosc. 61, 514–522 (2007).
(19) S. Glassford, K.L.A. Chan, B. Byrne, and S.G. Kazarian, Langmuir 28, 3174–3179 (2012).
(20) J.A. Kimber, S.G. Kazarian, and F. Štepánek, Powder Technol. 236, 179–187 (2013).
(21) K.L.A. Chan and S.G. Kazarian, Appl. Spectrosc. 57, 381–389 (2003).
(22) J. van der Weerd, K.L.A. Chan, and S.G. Kazarian, Vib. Spectrosc. 35, 9–13 (2004).
(23) O.S. Fleming and S.G. Kazarian, in Supercritical Carbon Dioxide, (Wiley-VCH Verlag GmbH & Co. KGaA, 2006), pp. 205–238.
(24) F.H. Tay and S.G. Kazarian, Energ. Fuel. 23, 4059–4067 (2009).
(25) T. Frosch, K.L.A. Chan, H.C. Wong, J.T. Cabral, and S.G. Kazarian, Langmuir. 26, 19027–19032 (2010).
(26) J. Pajander, S. Baldursdottir, J. Rantanen, and J. Østergaard, Int. J. Pharm. 427, 345–353 (2012).
(27) J. Østergaard, J.X. Wu, K. Naelapää, J.P. Boetker, H. Jensen, and J. Rantanen, J. Pharm Sci. 103, 1149–1156 (2014).
(28) C.A. Fyfe, H. Grondey, A.I. Blazek-Welsh, S.K. Chopra, and B.J. Fahie, J. Control Release. 68, 73–83 (2000).
(29) P. Kulinowski, A. Mlynarczyk, P. Dorozynski, K. Jasinski, M.H. Gruwel, B. Tomanek, and W. Weglarz, Pharm. Res. 29, 3420–3433 (2012).
(30) C.A. Coutts-Lendon, N.A. Wright, E.V. Mieso, and J.L. Koenig, J. Control Release 93, 223–248 (2003).
(31) H. Keles, A. Naylor, F. Clegg, and C. Sammon, Vib. Spectrosc. 71, 76–84 (2014).
(32) S. Kazarian and J. van der Weerd, Pharm. Res. 25, 853–860 (2008).
(33) S.G. Kazarian and A.V. Ewing, Expert Opin. Drug Del. 10, 1207–1221 (2013).
(34) J. van der Weerd, K.L. Andrew Chan, and S.G. Kazarian, Vib. Spectrosc. 35, 9–13 (2004).
(35) J.A. Kimber, S.G. Kazarian, and F. Štepánek, Comput. Chem. Eng. 35, 1328–1339 (2011).
(36) B. Zheng, L.S. Roach, and R.F. Ismagilov, J. Am. Chem. Soc. 125, 11170–11171 (2003).
(37) K. Ataka and J. Heberle, Anal. Bioanal. Chem. 388, 47–54 (2007).
(38) S.G. Kazarian, Macromol. Symp. 184, 215–228 (2002).
(39) S.G. Kazarian, Polym. Sc.i Ser. C+ 42, 78–101 (2000).
(40) S.G. Kazarian and K.L.A. Chan, Macromolecules 37, 579–584 (2004).
(41) A.P. Watkinson, Heat Transfer Eng. 28, 177–184 (2007).
(42) J.B. Cooper, K.L. Wise, W.T. Welch, M.B. Sumner, B.K. Wilt, and R.R. Bledsoe, Appl. Spectrosc. 51, 1613–1620 (1997).
(43) A.A. Gabrienko, C.H. Lai, and S.G. Kazarian, Energ. Fuel 28, 964–971 (2014).
(44) A. Gabrienko, V. Subramani, O. Martyanov, and S. Kazarian, Adsorpt. Sci. Technol. 32, 243–256 (2014).
(45) A.E. Pomerantz, D.J. Seifert, K.D. Bake, P.R. Craddock, O.C. Mullins, B.G. Kodalen, S. Mitra-Kirtley, and T.B. Bolin, Energ. Fuel. 27, 4604–4608 (2013).
(46) T. Tveter, P. Klaeboe, and C.J. Nielsen, Spectrochim. Acta A-M. 40, 351–359 (1984).
(47) K.L.A. Chan and S.G. Kazarian, Spectroscopy 27, 22–30 (2012).
(48) G.M. Whitesides, Nature 442, 368–373 (2006).
(49) K.L.A. Chan, X. Niu, A.J. de Mello, and S.G. Kazarian, Lab Chip 10, 2170–2174 (2010).
(50) K.L.A. Chan and S.G. Kazarian, Anal. Chem. 84, 4052–4056 (2012).
James A. Kimber, PhD, is a research associate and Sergei G. Kazarian is a professor of physical chemistry with the Department of Chemical Engineering at Imperial College London in London, England. Direct correspondence to: s.kazarian@imperial.ac.uk
Best of the Week: EAS Conference Coverage, IR Spectroscopy, Microplastics
November 22nd 2024Top articles published this week include highlights from the Eastern Analytical Symposium, a news article about the infrared (IR) spectroscopy market, and a couple of news articles recapping spectroscopic analysis of microplastics.
FT-IR Analysis of pH and Xylitol Driven Conformational Changes of Ovalbumin–Amide VI Band Study
November 21st 2024This study uses Fourier transform infrared (FT-IR) spectroscopy to analyze how the globular protein ovalbumin's secondary structures transition under varying pH conditions in the presence of the cosolvent xylitol, highlighting the role of noncovalent interactions in these conformational changes.
