Affinity Selection-Mass Spectrometry: Defining the Bioactive Compounds in Complex Mixtures of Natural Products and Combinatorial Libraries
Special Issues
Drug discovery using high-throughput screening of discreet compounds, and the discovery of natural products with pharmacological mechanisms of action, rely on bioassay-guided fractionation analysis. Recent applications of affinity selection–mass spectrometry (AS-MS) are useful for exploring the discovery of ligands to membrane-bound proteins and RNA targets.
Drug discovery from combinatorial libraries typically utilizes high-throughput screening of discreet compounds, and the discovery of natural products with pharmacological mechanisms of action relies on bioassay-guided fractionation. Both processes can be expedited through the application of affinity selection-mass spectrometry (AS-MS). AS-MS includes a family of MS-based affinity screening methods, including pulsed ultrafiltration (PUF)-AS-MS, size exclusion chromatography AS-MS, and magnetic microbead affinity selection screening (MagMASS). All AS-MS approaches begin by incubating a pharmacologically important receptor with a mixture of possible ligands, separating the ligand-receptor complexes from non-binding molecules (the approaches differ in this separation step), and then using LC–MS to characterize the affinity-extracted ligands. The speed, selectivity, and sensitivity of mass spectrometry and ultrahigh-pressure liquid chromatography (UHPLC)-compatible MS ionization techniques, like electrospray and atmospheric pressure chemical ionization, make AS-MS ideal for characterizing ligands. Recent applications of AS-MS include allosteric as well as orthosteric ligand discovery, and finding ligands to membrane-bound proteins and RNA targets.
The development of combinatorial chemistry in the 1990s facilitated the rapid synthesis of large numbers of drug-like molecules, called combinatorial libraries, for evaluation in pharmacological assays. The subsequent demand for faster and less expensive assays drove the development of high-throughput screening approaches, usually involving multiwall plate technologies, which enabled the screening of more than 10,000 compounds per day. Today, most high-throughput screening approaches test one compound at a time using rapid assays based on absorption or fluorescence such as fluorescence polarization (1), but growing in popularity are higher-throughput mass spectrometry (MS)-based screening approaches that screen combinatorial library mixtures.
By relying on mass selectivity, MS-based screening eliminates the need for radiolabels or chromophores. In particular, false positive results that can be caused by test compounds that exhibit interfering absorbance or fluorescence are avoided. This advantage is particularly important when screening natural products, which often contain strong chromophores. The need for more chemical diversity in screening programs than is available from combinatorial libraries has also renewed interests in natural products as sources of diverse chemical structures. In addition, since mass spectrometry-based assays do not require advance knowledge of the identities of the compounds in a mixture, these screening methods are ideal for natural products. Although the utility of mass spectrometry for natural products drug discovery was recognized at least 20 years ago (2), most
MS-based drug discovery assays used in the pharmaceutical industry remain focused on combinatorial
library screening (3).
The discovery of natural products with specific pharmacological mechanisms of action has been labor intensive, time consuming, and not easily adapted to a high-throughput formats. The standard approach today is bioassay-guided fractionation (4), which begins with a pharmacologically active natural product mixture such as an extract of a plant, fungus, or a microbial culture. The extract is partitioned with solvents, and the active partition is fractionated using chromatography. After assaying each fraction, the active fraction is subfractionated and re-assayed, until active compounds are isolated for spectroscopic analysis and identification. The application of mass spectrometry-based screening can expedite this process by condensing the reiterative fractionation/bioassay process into a single step. Because these mass spectrometry-based screening assays share a common affinity separation step (5), they have become known as affinity selection-mass spectrometry (AS-MS).
Affinity Selection-Mass Spectrometry (AS-MS)
A variety of mass spectrometry-based screening methods have been invented based on the affinity of active compounds for a pharmacological target. All of these methods utilize affinity interaction (the binding of a ligand to a receptor) to facilitate the isolation of active compounds from complex mixtures, and then MS to characterize the affinity-extracted ligands. The most popular and successful of these methods include size-exclusion chromatography (SEC)-AS-MS (6), pulsed ultrafiltration (PUF)-AS-MS (7), and magnetic microbead affinity selection MS (MagMASS) (8). In addition to the speed, selectivity, and sensitivity of mass spectrometry, the availability of LC-compatible MS ionization techniques like electrospray and atmospheric pressure chemical ionization have enabled these AS-MS methods to characterize a wide variety of ligands to receptors and enzymes. Other AS-MS approaches that have not yet been so widely adopted include affinity chromatography-MS (9), affinity capillary electrophoresis-MS (10), frontal affinity chromatography-MS (11), direct affinity screening of ligand-receptor complexes using electrospray ionization and ultrahigh resolution MS (12), and matrix-assisted laser desorption MS of ligands affinity-captured by an immobilized target (13).
Size exclusion Chromatography (SEC)-AS-MS
During SEC-AS-MS, a mixture of compounds is incubated with a pharmacological receptor and allowed to come to equilibrium (Figure 1). Any binding buffer may be used, including non-volatile buffers that are incompatible with mass spectrometry, since the buffer will be removed during the final stage of reverse phase LC–MS analysis. The ligand-receptor complex, which is much larger than the unbound ligands, is separated from the low mass, non-binding compounds using SEC (14). Also known as gel permeation chromatography, SEC separates molecules according to size as they pass through a stationary phase containing particles with pores that allow small molecules to enter but exclude large molecules. The large ligand-receptor complexes elute first.
Figure 1: Comparison ofFigure 1: Comparison of SEC-AS-MS and PUF-AS-MS screening. After incubation of a receptor with a mixture of compounds, the large ligand–receptor complexes are separated from the small unbound compounds using either size exclusion chromatography (SEC) or ultrafiltration. The isolated ligand-receptor complexes are denatured to release the ligands either off-line or during the final UHPLC–MS/MS analysis step.
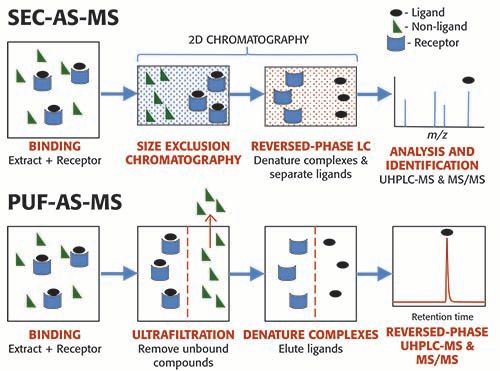
The ligands are released from the receptor using organic solvent or a pH change that denatures the receptor. This denaturing step can be carried out prior to LC–MS analysis, or directly on the reversed-phase column. For maximum sensitivity, 2-dimensional SEC-reversed-phase chromatography is used, in which the ligand-receptor complexes eluting from the SEC column are directed on-line onto the reversed -phase column for the final LC–MS or UHPLC–MS step. Besides serving to separate the ligands from each other and from the receptor, the reversed-phase chromatographic separation also removes the binding buffer, which is often incompatible with mass spectrometric analysis. The SEC separation must be carried out quickly and at reduced temperatures (usually 4 °C), because ligands begin to dissociate from the receptor immediately and can become lost into the size exclusion stationary phase.
SEC-LC–MS was invented by Kaur and co-workers (15), and was originally used to screen peptide libraries for ligands of a protein target. Since then, SEC-AS-MS been automated to include 2-D chromatography (SEC-reversed-phase) and has been applied to a wide variety of targets (16,17). In this way, pharmaceutical researchers can screen over 100,000 combinatorial library compounds per day, in mixtures routinely containing up to 2,600 compounds at a time (18,19). In natural product applications, SEC-AS-MS has been used to identify pharmacologically active compounds in complex botanical extracts such as traditional Chinese medicines (20,21). Some of the advantages and disadvantages of SEC-AS-MS are summarized in Table I.
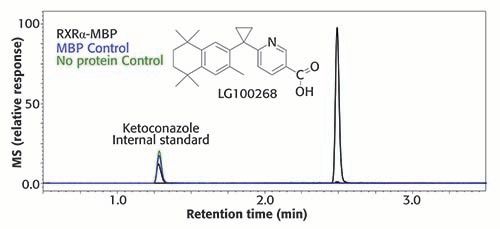
As an example of SEC-AS-MS data, reversed-phase LC–MS chromatograms for the screening for ligands of the nuclear receptor, retinoid X receptor (RXRα), are shown in Figure 2. RXRα is under investigation as a drug discovery target for anti-inflammation therapy as well as for cancer treatment and prevention. The chromatograms represent two negative control incubations and a positive control. Note the intense chromatographic peak for the known ligand LG100268 in the positive control, and almost no signal or background for the negative control incubations. The background noise of SEC-AS-MS assays is characteristically lower than other AS-MS screening approaches. In this example, UHPLC–MS was used instead of HPLC for faster separations (Figure 2).
Figure 2: SEC-AS-MS screening for ligands of retinoid X receptor (RXR)-α expressed recombinantly with a maltose binding protein (MBP) tag. The known ligand LG100268 (used as a positive control) was detected in the assay using RXRα-MBP but not in the controls using MBP alone or no protein. Ketoconazole was added after the SEC separation as an internal standard to normalize and compare the reversed phase UHPLC–MS chromatograms.
Pulsed Ultrafiltration (PUF)-AS-MS
PUF-AS-MS screening begins with the incubation of a mixture of potential ligands with a macromolecular target in solution, such as an enzyme or receptor. The ligand-receptor complexes are then separated from the unbound compounds using ultrafiltration. As with SEC-AS-MS, the ultrafiltration separation of bound ligands from unbound compounds should be carried out quickly, and at reduced temperature, to minimize dissociation and loss of ligand. The ligand-receptor complexes are denatured, usually using water and acetonitrile acidified with a volatile organic acid such as formic acid, and the released ligands are trapped on a reversed-phase HPLC or UHPLC column for characterization using (UHPLC–MS and MS/MS (Figure 1) (22).
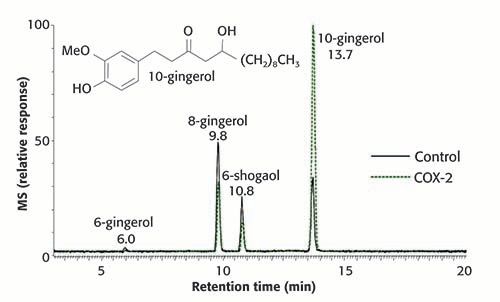
As a natural products application of PUF-AS-MS, an extract of ginger (Zingiber officinale Roscoe) was screened for ligands of the inflammation target cyclooxygenase-2 (COX-2) (23) (Figure 3). Ginger roots have been used traditionally to treat inflammation, and had been reported to inhibit COX. To identify the pharmacologically active compounds in ginger, PUF-AS-MS was used to screen a chloroform partition of a methanol extract of ginger roots for COX-2 ligands. A series of gingerols and shogaols were determined using PUF-AS-MS to bind to COX-2 (Figure 3). Competitive binding experiments using PUF-AS-MS with ginger and the known synthetic inhibitor celecoxib indicated that the ginger ligands bound to the active site of COX-2. Additional functional enzyme assays confirmed that these ginger compounds inhibited COX-2 but not COX-1 with IC-50 values in the low micromolar range.
Figure 3: PUF-MS screening of an extract of ginger for ligands to the inflammation target cyclooxygenase-2 (COX-2). After pulsed ultrafiltration, the ultrafiltrates of experiment (incubations with functional COX-2) and control (incubation without receptor) were analyzed using negative ion electrospray mass spectrometry with reversed phase HPLC separation. Gingerols and shogaols were detected as COX-2 ligands based on enhancement of the LC–MS signal relative to the control. (Adapted from van Breemen, et al. [23] and used with permission.)
Invented in the van Breemen laboratory in the 1990s (24), PUF -AS-MS has been adopted for use by both academic (25,26) and industrial researchers (27) to screen combinatorial libraries in pools of up to 2,700 compounds each (28), as well as natural product extracts (29, 30) for the discovery of ligands to a wide variety of macromolecular receptors (31,32). Advantages and disadvantages of PUF-AS-MS are listed in Table I
Magnetic Microbead Affinity Selection Screening (MagMASS)
A newer AS-MS method, MagMASS was first reported in 2008 by Choi and van Breemen (33) for screening combinatorial library mixtures and natural product extracts for ligands to an immobilized target. Recently, MagMASS became the first AS-MS approach to incorporate multititer well plates, which facilitated automation and enhanced the throughput (8,34). MagMASS has also been applied to the screening of complex botanical extracts (34).
The MagMASS process (Figure 4) begins by immobilizing a pharmacological receptor to magnetic beads either covalently or non-covalently. A variety of bead surface chemistries are available from commercial sources for covalently immobilizing receptors. Non-covalent immobilization is also possible by utilizing interactions such as immobilized streptavidin binding biotinylated receptors, immobilized nickel ions binding with receptors containing His-tags, and immobilized amylose interacting with receptors containing maltose binding protein.
Figure 4: For MagMASS, the
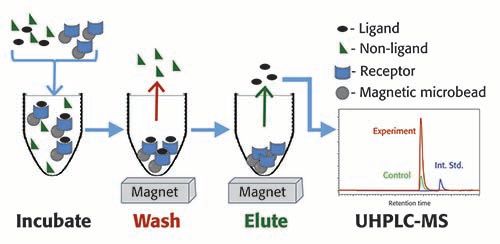
After immobilization of the receptor, the magnetic microbeads are incubated with mixtures of potential ligands (Figure 4). A washing step is used to remove the unbound compounds while a magnetic field retains the beads containing the receptor-ligand complexes. Ligands are then released using a denaturing solvent or a pH change for analysis using (UHPLC–MS and (UHPLC–MS/MS. To control for non-specific binding, beads without receptor are usually incubated in parallel. See Table I for a summary of advantages and disadvantages of MagMASS and comparison with SEC-AS-MS and PUF-AS-MS.
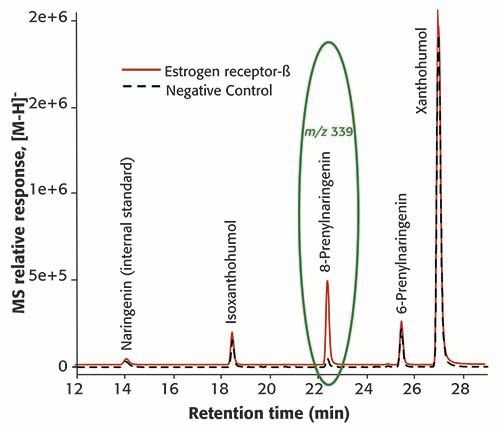
As an example of applying MagMASS to the screening of complex botanical extracts, human estrogen receptor-β was immobilized covalently on aldehyde functionalized magnetic beads and used to screen for botanical estrogens (Figure 5) (33). Following incubation with extracts of hops (Humulus lupulus L.), the non-binding compounds were washed away using buffer, and then bound ligands were released by denaturing the protein with methanol. The natural product 8-prenylnaringenin was identified as the most active estrogen in hops (Figure 5).
Figure 5: An extract of hops (Humulus lupulus L.) was screened for ligands to the human estrogen receptor-β (ER-β) using MagMASS with positive ion electrospray LC–MS. The prenylated flavonone 8-prenylnaringenin was identified as the most potent estrogen in the extract. (Adapted from Choi and van Breemen [33] and used with permission.)
AS-MS Data Analysis
For all AS-MS approaches, high resolution tandem mass spectrometers are preferred, such as quadrupole time-of-flight or orbitrap mass spectrometers, so that elemental compositions of ligands may be determined. When screening combinatorial libraries, the masses and elemental compositions of each compound are already known and may be entered into a database. By using high resolution accurate mass measurements during screening, the elemental compositions and isotope patterns of ligands may be compared with the database either manually or automatically using commercially available metabolomics software. For additional confirmation of ligand identity and for distinguishing among isomeric compounds, tandem mass spectra may be compared with those of standards.
The identification of ligands from natural product extracts is more challenging, but also begins with high resolution MS and MS/MS analyses to determine elemental composition and structural features. By comparing the measured elemental compositions and tandem mass spectra with those of known natural products in databases in a process known as dereplication, ligand structures may be determined quickly. Reversed-phase UHPLC retention times, obtained during screening step, enable novel natural product ligands to be collected for additional structure determination using spectroscopic techniques such as NMR.
Comparison of AS-MS Approaches and Novel Applications
Advantages of all AS-MS methods include the diversity of receptors that may be screened, the ability to screen complex natural product extracts as well as combinatorial libraries, compatibility with any incubation buffer, low consumption of receptors and cofactors, elimination of radiolabels and chromophores, lack of interference from matrix, and lower costs and faster screening than the current standard of screening discreet compounds (Table I). Due to the procedural step of separating ligand-receptor complexes from unbound compounds, all AS-MS methods share the common disadvantage of reduced sensitivity for rapidly dissociating ligands. The reversed-phase chromatography separation is a relatively slow step in the AS-MS process, but this step has been accelerated by implementing UHPLC in place of HPLC.
Although SEC-AS-MS and PUF- AS-MS enable solution-phase screening, MagMASS requires receptor immobilization, which has the potential disadvantage of altering binding properties (Table I). On the other hand, receptor immobilization for MagMASS can facilitate the screening of membrane-bound or poorly soluble receptors. In an online configuration of PUF-AS-MS that uses MS-compatible buffers without column chromatography, PUF-AS-MS has the advantage of enabling the measurement of affinity constants, stoichiometry of binding, and enzyme kinetics for ligand–receptor interactions (35,36).
AS-MS offers substantial improvements in speed and convenience over alternative screening approaches, and many novel applications have been reported just recently (Table I). For example, AS-MS may be used to screen an equimolar mixture of natural products (31) or combinatorial library compounds (37) and, in a single experiment, rank order the ligands in order of affinity for the receptor. Note that the conventional approach to ranking ligands for binding affinity requires individual assays of each compound for competition with a radiolabeled ligand. Unlike competition binding assays, AS-MS may be used to discover ligands that bind to allosteric sites (37,38), and provide lead compounds for targets previously considered “undruggable” (39). Use of enzymes and receptors embedded in microsomes (40) or cell membranes (41,42) has also been demonstrated. AS-MS is not limited to screening for ligand to protein receptors, as other macromolecular targets such as RNA molecules have been used (43,44).
Conclusions
In addition to providing a faster and less expensive alternative to conventional screening of discreet compounds for interactions with receptors, AS-MS addresses the important and yet unmet need for technology that facilitates the rapid discovery of natural products with pharmacological activities from complex mixtures such as botanical and microbial extracts. By eliminating the need for spectroscopic detection, AS-MS eliminates many limitations of conventional high-throughput screening, such as matrix interference or false positives due to strong absorbance or fluorescence of test compounds. In addition, AS-MS offers a variety or unique applications that are not easily addressed or have not been achieved using conventional high-throughput screening such as discovering allosteric ligands, ligands to RNA, and ligands to membrane-bound receptors. Although invented over 20 years ago (15,24), many of the unique applications of AS-MS have only recently been demonstrated. Therefore, expect many more innovations and natural products applications of AS-MS during the next 20 years.
References
- W.A. Lea, and A. Simeonov, Expert Opin. Drug Discov. 6, 17–32 (2011).
- J. Liu, J.E. Burdette, H. Xu, C. Gu, R.B. van Breemen, K.P. Bhat, N. Booth, A.I. Constantinou, J.M. Pezzuto, H.H. Fong, N.R. Farnsworth, and J.L. Bolton, J. Agric. Food Chem.49, 2472–2479 (2001).
- S.S. Walker, D. Degen, E. Nickbarg, D. Carr, A. Soriano, M. Mandal, R.E. Painter, P. Sheth, L. Xiao, X. Sher, N. Murgolo, J. Su, D.B. Olsen, R.H. Ebright, and K. Young, ACS Chem. Biol.12, 1346–1352 (2017).
- M.G. Weller, Sensors (Basel)12, 9181–9209 (2012).
- Y.G. Shin, and R.B. van Breemen, Biopharmaceut. Drug Dispos. 22, 353–372 (2001).
- F. Touti, Z.P. Gates, A. Bandyopdhyay, G. Lautrette, and B.L. Pentelute, Nat. Chem. Biol. 15, 410–418 (2019).
- B.M. Johnson, D. Nikolic, and R.B. van Breemen, Mass Spectrom. Rev. 21, 76-86 (2002).
- M.D. Rush, E.M. Walker, G. Prehna, T. Burton, and R.B. van Breemen, J. Am. Soc. Mass Spectrom.28, 479–448 (2017).
- M.A. Kelly, H. Liang, I.I. Sytwu, I. Vlattas, N.L. Lyons, B.R. Bowen, and L.P. Wennogle, Biochemistry35, 11747–11755 (1996).
- Y.-H. Chu, L.Z. Avila, J. Gao, and G.M. Whitesides, Ace. Chem. Res. 28, 461–468 (1995).
- E.S.M. Ng, F. Yang, A. Kameyama, M.M. Palcic, O. Hindsgaul, and D.C. Schriemer, Anal. Chem. 77, 6125–6133 (2005).
- H. Vu, N.B. Pham, and R.J. Quinn, Biomol. Screen.13, 265–275 (2008).
- E.C. VanderPorten, M.D. Scholle, J. Sherrill, J.C. Tran, and Y. Liu, SLAS Discov.22, 1211–1217 (2017).
- S. Mathur, J.D. Park, D.H. Kim, and R.W. Hartmann. J. Biomol. Screen. 10, 30–35 (2005).
- S. Kaur, L. McGuire, D. Tang, G. Dollinger, and V. Huebner, J. Protein Chem. 16, 505–511 (1997).
- T.N. O’Connell, J. Ramsay, S.F. Rieth, M.J. Shapiro, and J.G. Stroh. Anal. Chem. 86, 7413–7420 (2014).
- V.D. Kutilek, C.L. Andrews, M.P. Richards, Z. Xu, T. Sun T, Y. Chen, A. Hashke, N. Smotrov, R. Fernandez, E.B. Nickbarg, C. Chamberlin, B. Sauvagnat, P.J. Curran, R. Boinay, P. Saradjian, S.J. Allen, N. Byrne, N.L. Elsen, R.E. Ford, D.L. Hall, M. Kornienko, K.W. Rickert, S. Sharma, J.M. Shipman, K.J. Lumb, K. Coleman, P.J. Dandliker, I. Kariv, and B. Beutel, J. Biomol. Screen.21, 608–619 (2016).
- Y. Deng, G.W. Shipps, Jr, L. Zhao, M.A. Siddiqui, J. Popovici-Muller, P.J. Curran, J.S. Duca, A.W. Hruza, T.O. Fischmann, V.S. Madison, R. Zhang, C.W. McNemar, T.W. Mayhood, R. Syto, A. Annis, P. Kirschmeier, E.M. Lees, D.A. Parry, and W.T. Windsor, Bioorg. Med. Chem. Lett.24, 199–203 (2014).
- X. Huang, C.C. Cheng, T.O. Fischmann, J.S. Duca, X. Yang, M. Richards, and G.W. Shipps, Jr., ACS Med. Chem. Lett. 3, 123–128 (2012).
- Y. Fu, H.Y. Mo, W. Gao, J.Y. Hong, J. Lu, P. Li, and J. Chen, Anal. Bioanal. Chem. 406, 4987–4995 (2014).
- F. Fei, H. Sun, X. Cheng, J. Liu, J. Wang, Q. Li, and Y. Zhang, Chin. Med. 13, 49 (2018).
- B.M. Johnson, D. Nikolic, and R.B. van Breemen, Mass Spectrom. Rev.21, 76–86 (2002).
- R.B. van Breemen, Y. Tao, and W. Li, Fitoterapia82, 38–43 (2011).
- R.B. van Breemen, C.H. Huang, D. Nikolic, C.P. Woodbury, Y.Z. Zhao, and D.L. Venton, Anal. Chem. 69, 2159–2164 (1997). US Patent No. US 5872015 (filed May 10, 1996)
- R. Munigunti, V. Mulabagal, and A.I. Calderón, J. Pharm. Biomed. Anal.55, 265–271 (2011).
- J.L. Zhou, Z.M. Qian, Y.D. Luo, D. Tang, H. Chen, L. Yi, and P. Li, Biomed. Chromatogr.22, 1164–1172 (2008).
- M.B. Beverly, P. West, and R.K. Julian, Comb. Chem. High Throughput Screen. 5, 65–73 (2002).
- E.K. Comess, J.E. Schurdak, M.J. Voorbach, M. Coen, J.D. Trumbull, H. Yang, L. Gao, H. Tang, X. Cheng, C.G. Lerner, J.O. McCall, D.J. Burns, and B.A. Beutel, J. Biomol. Screen. 11, 743–754 (2006).
- H. Li, F. Song, J. Xing, R. Tsao, Z. Liu, and S. Liu, J. Am. Soc. Mass Spectrom. 20, 1496–1503 (2009).
- Y. Choi, Y. Jung, and S.N. Kim, Molecules 20, 13753–13763 (2015).
- Y. Sun, C. Gu, X. Liu, W. Liang, P. Yao, J.L. Bolton, and R.B. van Breemen, J. Am. Soc. Mass Spectrom.16, 271–279 (2005).
- D. Liu, J. Guo, Y. Luo, D.J. Broderick, M.I. Schimerlik, J.M. Pezzuto, and R.B. van Breemen, Anal. Chem.79, 9398–9402 (2007).
- Y. Choi, and R.B. van Breemen, Comb. Chem. High Throughput Screen.11, 1–6 (2008).
- M.D. Rush, E.M. Walker, T. Burton, and R.B. van Breemen, J. Nat. Prod. 79, 2898–2902 (2016).
- C. Gu, D. Nikolic, J. Lai, X. Xu, and R.B. van Breemen, Combin. Chem. High Throughput Screen.2, 353–359 (1999).
- B.M. Johnson, D. Nikolic, and R.B. van Breemen, Mass Spectrom. Rev. 21, 76–86 (2002).
- D.A. Annis, N. Nazef, C.C. Chuang, M.P. Scott, and H.M. Nash, J. Am. Chem. Soc.126, 15495–15503 (2004).
- C.E. Whitehurst, N. Nazef, D.A. Annis, Y. Hou, D.M. Murphy, P. Spacciapoli, Z. Yao, M.R. Ziebell, C.C. Cheng, G.W. Shipps Jr, J.S. Felsch, D. Lau, and H.M. Nash, J. Biomol. Screen.11, 194–207 (2006).
- W.L. Petrilli, G.C. Adam, R.S. Erdmann, P. Abeywickrema, V. Agnani, X. Ai, J. Baysarowich, N. Byrne, J.P. Caldwell, W. Chang, E. DiNunzio, Z. Feng, R. Ford, S. Ha, Y. Huang, B. Hubbard, J.M. Johnston, M. Kavana, J.-M. Lisnock, R. Liang, J. Lu, Z. Lu, J. Meng, P. Orth, O. Palyha, G. Parthasarathy, S.P. Salowe, S. Sharma, J. Shipman, S.M. Soisson, A.M. Strack, H. Youm, K. Zhao, D.L. Zink, H. Zokian, G.H. Addona, K. Akinsanya, J.R. Tata, Y. Xiong, and J.E. Imbriglio, Cell Chem. Biol.26, 32–40 (2020).
- Y.G. Shin, J.L. Bolton, and R.B. van Breemen, Comb. Chem. High Throughput Screen.5, 59–64 (2002).
- Y. Lu, S. Qin, B. Zhang, A. Dai, X. Cai, M. Ma, Z.-G. Gao, D. Yang, R.C. Stevens, K.A. Jacobson, M.-W. Wang, and W. Shui, Anal. Chem.91, 8162–8169 (2019).
- S. Qin, M. Meng, D. Yang, W. Bai, Y. Lu, Y. Peng, G. Song, Y. Wu, Q. Zhou, S. Zhao, X. Huang, J.D. McCorvy, X. Cai, A. Dai, B.L. Roth, M.A. Hanson, Z.-J. Liu, M.-W. Wang, R.C. Stevens, and W. Shui, Chem. Sci. 9, 3192–3199 (2018).
- N.F. Rizvi, and E.B. Nickbarg, Methods 167, 28–38 (2019).
- N.F. Rizvi, J.A. Howe, A. Nahvi, D.J. Klein, T.O. Fischmann, H.-Y. Kim, M.A. McCoy, S.S. Walker, A. Hruza, M.P. Richards, C. Chamberlin, P. Saradjian, M.T. Butko, G. Mercado, J. Burchard, C. Strickland, P.J. Dandliker, G.F. Smith, and E.B. Nickbarg, ACS Chem. Biol. 13, 820–831 (2018)
Richard van Breemen is with the Linus Pauling Institute at Oregon State University in Corvallis, Oregon. Direct correspondence to: richard.vanbreemen@oregonstate.edu

High-Speed Laser MS for Precise, Prep-Free Environmental Particle Tracking
April 21st 2025Scientists at Oak Ridge National Laboratory have demonstrated that a fast, laser-based mass spectrometry method—LA-ICP-TOF-MS—can accurately detect and identify airborne environmental particles, including toxic metal particles like ruthenium, without the need for complex sample preparation. The work offers a breakthrough in rapid, high-resolution analysis of environmental pollutants.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)








