XRF in Agronomy Applications—Analysis of Plant Tissues and Fertilizers
Special Issues
New excitation developments and advanced detector technology enable the use of EDXRF for multielement analysis of plant material and fertilizers, with improved detection limits and reduced measurement time. These features are combined with easy sample preparation and low cost of investment.
For the elemental analysis of plant tissue materials, traditionally ICP-OES or ICP-MS are used. The advantages are clear, as the sample prep by digestion is standardized. The instruments deliver the required sensitivity and precision, and the measurement itself is very fast and can be automated. The same is true when analyzing fertilizer samples. However, the infrastructure necessary to operate an ICP instrument is not always available, or possible. An additional drawback of the traditional approaches is the chemical waste, resulting from the acids required for digestion. For fast on-site analysis of plant materials, portable energy-dispersive XRF (EDXRF) is a very suitable tool. For applications in the lab demanding high precision and lower detection limits, EDXRF in the past was limited, as the measurement times needed for the required precision were too long, and detection limits for some critical trace elements were not low enough. Based on new excitation concepts and advanced detector technology, modern EDXRF instruments are suitable to perform plant tissue analysis within a much shorter measurement time. Also, highly accurate analyses of major and minor elements in fertilizers can be done. For some critical trace elements in plant tissue and fertilizers, excellent detection limits can be achieved. This is combined with easy sample preparation and low investment into the infrastructure needed to operate the instrument.
The determination of plant nutrients, trace elements, and heavy metals in plant tissue and in fertilizers is of great importance. High crop yields require a sufficient supply of plant nutrients, such as phosphorus and potassium, and of essential trace elements, such as copper and zinc. At the same time, the content of possibly harmful, heavy metals, such as lead, cadmium, nickel, arsenic, and uranium, must be kept low, in order to avoid accumulation in soil and their entrance into the food chain.
In December 2018, the European Union decided to introduce limits for toxic contaminants in certain fertilizers, including a 60 mg/kg limit for cadmium. To limit the pick-up of uranium by plants, the German Environment Agency recommends a maximum concentration of 50 mg/kg of phosphate.
XRF allows determination of potassium and phosphorus concentration in fertilizer. It also is suitable as a fast screening tool to test for trace element content. For example, important trace elements, are cadmium and uranium as possible contaminants of fertilizer.
Improvements in detector and digital signal processing techniques have opened new possibilities for multi-element analysis using EDXRF instruments. In combination with optimized excitation, high sensitivities and low background levels can be achieved. The instrument setup is described in (1). For the application example described in this article, the following benefits can be achieved:
The analysis of nutritional elements in plant tissue-this can accomplished with shorter measurement times.
Lower detection limits for critical trace elements can be realized.
Major element content in fertilizer material can be determined with high precision, which is a basic requirement for achieving high accuracy.
Materials and Methods
The analysis of plant materials for toxic and nutritional elements requires little sample preparation. The sample material was dried and pulverized. From the powdered material, 8 g were taken and pressed into a pellet with a diameter of 40 mm, using a pressure of 15 tons.
All measurements were made using a Spectro XEPOS simultaneous EDXRF spectrometer (Spectro Analytical Instruments). This instrument has an air-cooled, 50 W end-window X-ray tube. The tube anode consists of a thick, binary-alloy cobalt-palladium construction, combined with an adaptive excitation system. The analyzer includes a high-resolution silicon drift detector (SDD), plus the fixed excitation optics, and a filter changer in front of the X-ray tube. The resolution of the SDD is <130 eV (Mn Kα) at an input rate of 1 million counts per s (cps).
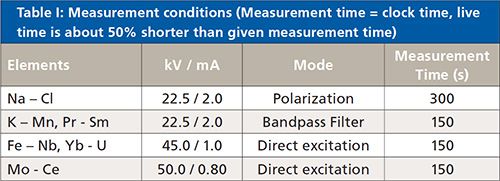
Table I lists the measurement conditions of the EDXRF instrument.
The analysis of fertilizer samples also requires drying and pulverization of the samples. The analysis of trace element content can be done from powdered samples. For sampling, 4 g of the fine powder is placed into a disposable sample cup with an outer diameter of 32 mm, which is typically closed with a polypropylene foil of 4 µm thickness at the analysis side. The main components, such as phosphorus pentoxide (P2O5), potassium oxide (K2O), and magnesium oxide (MgO), are ideally analyzed as fused beads. To prepare the fused beads, 1 g of the fine powdered material was mixed with 8 g of Lithium tetraborate and fused to a bead with 40 mm diameter using an automatic fluxer.
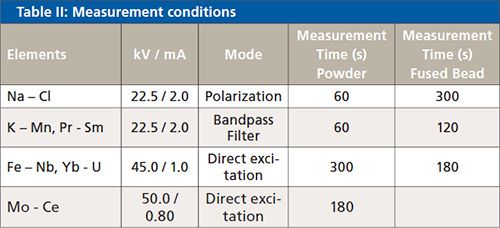
Table II lists the measurement conditions of the EDXRF instrument.
Results
Analysis of Plant Materials for Toxic and Nutritional Elements
For the analysis of plant tissue samples, the analyzer was calibrated using various well-characterized plant material samples. The calibration is based on the fundamental parameters approach. The validation of the method was done using two reference samples from WEPAL (2) and from NIST (3).
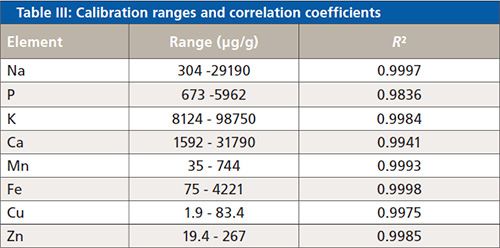
Table III lists the calibration ranges for some minor and trace elements as well as the correlation coefficients.
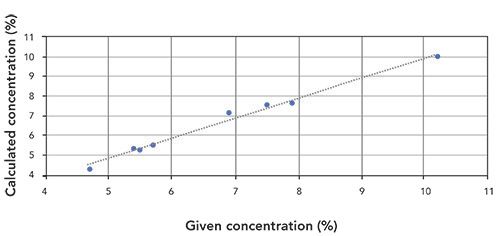
The following example correlation graphs for P (Figure 1) and K (Figure 2) show good agreement between the reference values and the analyzed concentrations.
Figure 1: Correlation graph for P content in plant tissue samples (R2 = 0.9836).
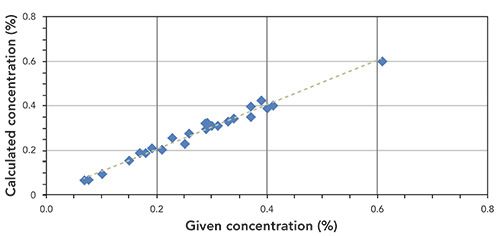
Figure 2: Correlation graph for K content in plant tissue samples (R2 = 0.9984).
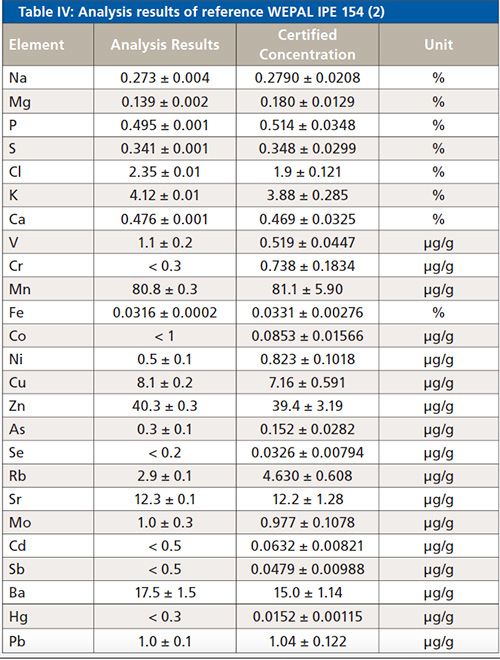
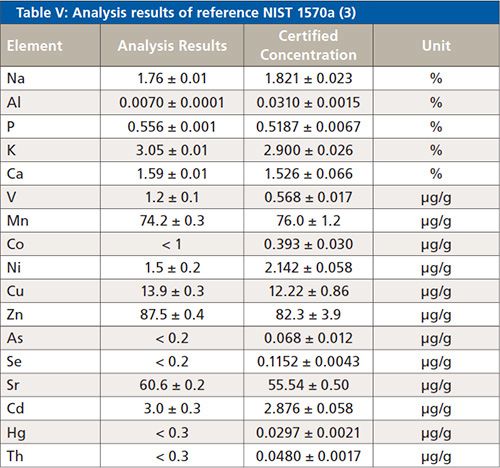
To validate the method, two reference samples were analyzed (Analysis Results). These results are displayed in Tables IV and V. Information values from the certificate (Certified Concentration) are printed in italics. The printed error of the analysis results is the statistical error for a 1 sigma confidence interval.
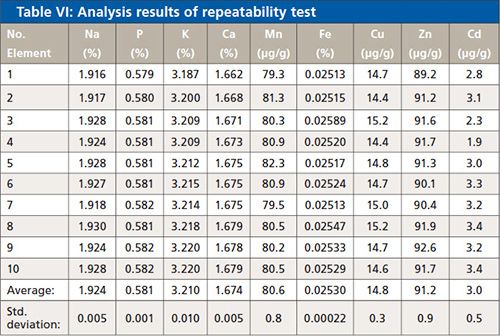
To test the precision of the EDXRF, the NIST 1570a reference sample was analyzed ten times. The results are displayed in Table VI.
Analysis of Fertilizers for Toxic and Nutritional Elements
For the analysis of the fused beads, the calibration was done using various well-characterized fertilizer samples. The calibration is based on the fundamental parameters approach.
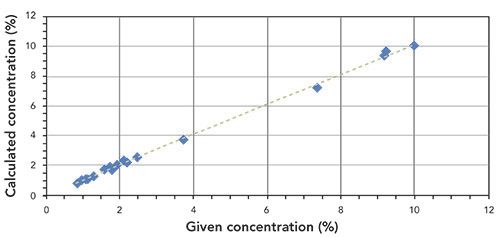
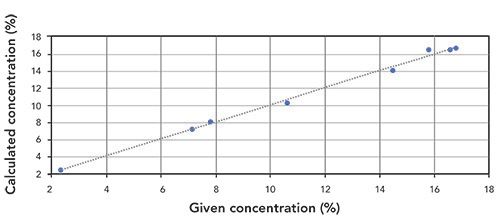
Figures 3 and 4 show the correlation graphs for the analysis of P2O5 and K2O in fertilizer.
Figure 4: Correlation graph for K2O content in fertilizer samples (R2 = 0.9957).
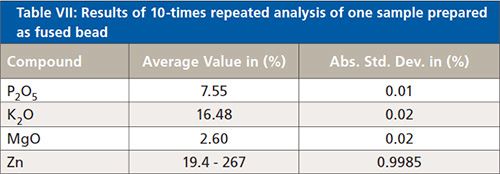
To test the precision of the analyzer, one of the fused bead samples was analyzed 10 times. The results are displayed in Table VII.
For the analysis of the powder samples, a screening method for the analysis of various types of geological samples was applied. This screening method is based on a combination of fundamental parameters for fluorescence and scattering and on a Compton-normalization approach.
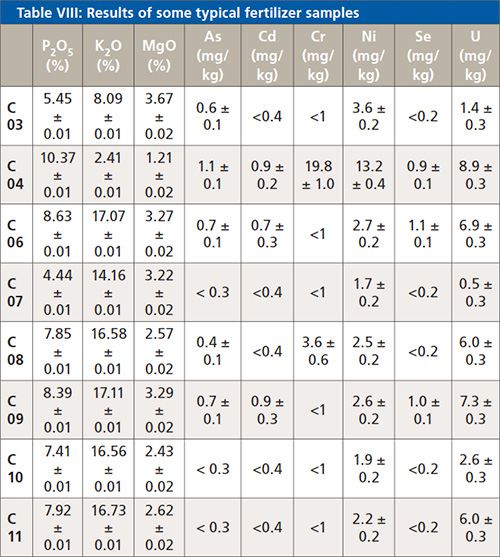
Table VIII shows a summary of some typical fertilizer samples. The results for the major elements were taken from the analysis of the fused beads, those results for the trace elements are taken from the analysis of the powder samples. The printed error is the statistical error for a 1 sigma confidence interval.
Summary and Conclusions
Since the sample preparation, especially when analyzing powder samples, involves only a little effort, an analysis using EDXRF can be done in a smaller lab, which is not equipped with a sample prep infrastructure required for ICP-OES or ICP-MS analyses.
The determination of the content of major and minor elements can be done with good precision. For labs running batches of high numbers of samples, the relatively long measurement times of XRF are a drawback compared to those using ICP techniques. Therefore, EDXRF is very suitable for labs with low to medium sample throughput.
The detection limits for some critical trace elements are sufficient for a quick screening analysis, but may not be sufficient for every application. Looking at the maximum tolerable limit of 50 milligrams uranium per kilogram of phosphate, as proposed by the German Federal Institute for Risk Assessment, this means a content of 2.5 to 5 mg/kg of uranium in the fertilizer. Reaching a limit of quantification of 2.5 mg/kg would mean reaching a limit of detection of <1 mg/kg, which is a challenge for XRF.
The improvements in detector and digital signal processing techniques have opened new possibilities for multi-element analysis of plant tissue and fertilizer samples using energy dispersive XRF. As described the technique enables good precision and low detection limits and can be recommended to be used in labs with low to medium sample throughput.
References
- J. Heckel and D. Wissmann, Spectroscopy 31(7),24–29 (2016).
- Certificate of Analysis for Reference Material IPE Sample 154, Raye Grass /Lolium perenne (Wageningen Evaluation Programs for Analytical Laboratories, Wageningen, The Netherlands)
- Certificate for SRM 1570a - Trace Elements in Spinach Leaves (National Institute of Standards and Technology, Washington, D.C., 2002) .
Additional Resources
R.E. Van Grieken and A.A. Markowicz, Eds., Handbook of X-ray Spectrometry (Marcel Dekker Inc., New York, New York, 2nd Ed.,1982).
J. Willis, C. Feather, and K. Turner, Guidelines for XRF Analysis (James Willis Consultants Cape Town, South Africa, 2014).
Dirk Wissmann is the Product Manager of XRF instruments with Spectro Analytical Instruments, in Kleve, Gemany. Direct correspondence to: dirk.wissmann@ametek.com

Trending on Spectroscopy: The Top Content of 2024
December 30th 2024In 2024, we launched multiple content series, covered major conferences, presented two awards, and continued our monthly Analytically Speaking episodes. Below, you'll find a selection of the most popular content from Spectroscopy over the past year.
Best of the Week: Hyperspectral Imaging, ICP-MS Analysis of Geological Samples, Product Roundup
October 18th 2024Top articles published this week include an article about hyperspectral imaging in human skin research, a peer-reviewed article about analyzing geological samples using atomic spectroscopy techniques, and an equipment roundup piece about the latest products in the industry.