Studying Gallstones and Kidney Stones with WDXRF
Special Issues
The accumulation of heavy and toxic elements in gallstones and kidney stones can be studied using WDXRF spectroscopy. Here, Vivek K. Singh of Shri Mata Vaishno Devi University in India talks about his work using this technique to understand the formation and content of these stones in the body.
Gallstones and kidney stones are very painful, and also can lead to cancer. The accumulation of heavy and toxic elements has been associated with the formation of such stones. Wavelength dispersive X-ray fluorescence (WDXRF) spectroscopy can be a useful technique for analyzing the types and concentrations of such elements in these stones as a means of better understanding their causes and development, as well as potential ways to avoid them. Vivek K. Singh, an assistant professor of physics at Shri Mata Vaishno Devi University in Katra, India, has been studying the use of WDXRF for such analyses, and he recently spoke to us about this work.
Laura Bush
You recently conducted a study of the use of wavelength dispersive X-ray fluorescence (WDXRF) spectroscopy for the detection and quantification of heavy metals in gallstones (1). First, why is such determination important? And what does information about the presence of heavy metals reveal?
Stone formations (such as gallstones and kidney stones) inside the human body are very painful diseases that also involve time and cost. Accumulation of heavy and toxic elements inside the various organs is one of the main causes of stone formation, which also leads to gallbladder cancer if it remains untreated. Detection and quantification of these elements within stones is important so as to define etiopathogenic factors that, in turn, are useful to identify preventive and therapeutic strategies. This is why we have started to investigate and quantify a wide range of heavy and toxic elements in different types of stones, employing advanced elemental analysis technology such as WDXRF. These elements were found responsible for the formation and growth of gallstones. Most importantly, these studies provide information about the dietary habits to be followed by concerned patients, which could be very beneficial to prevent stone formation and their recurrence.
Why did you want to evaluate the use of WDXRF for this type of analysis? Specifically, what advantages does it have compared to energy-dispersive XRF (EDXRF), as well as other elemental spectroscopy techniques, such as inductively coupled plasma–mass spectrometry (ICP-MS) or laser-induced breakdown spectroscopy (LIBS)?
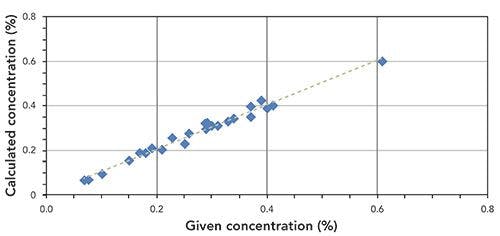
WDXRF spectroscopy is a modern technology that is capable of measuring all elements from beryllium (Be) to uranium (U) simultaneously in a concentration range from a few ppm to 100%, and can provide reliable and quality data. Compared with atomic absorption (AA) spectroscopy and ICP-MS, WDXRF is very fast and versatile. Techniques such as AA and ICP-MS are very time consuming, and can be hard to use because of sample preparation protocols in liquid form, which is very difficult in the case of gallstones and kidney stones. Also, the spatial distributions of the heavy elements are lost within the stones during sample dissolution. On the other hand, using micro-XRF, spatial information can also be obtained within the stones, which will be very useful to elucidate their growth and development processes. Although AA and ICP-MS are still very sensitive elemental analysis techniques, XRF has now become an advanced technology for bioscientists for multielemental research to analyze biomaterials, given its nondestructive nature. Compared with EDXRF, WDRXF provides high resolution to reduce spectral overlaps and thus more complex biosamples, such as stones, can be analyzed accurately. LIBS is a very strong competitor of XRF for elemental analysis. When using LIBS for the quantitative analysis of biological samples such as kidney stones, certified reference materials (CRMs) are required in order to obtain calibration curves, which are very difficult to obtain for heterogeneous kidney stones. In the present study using WDXRF, a specialized standardless software was used to quantify the elements. The EDXRF system and software can be used together to get very good results with biological samples, and we have used them together to analyze different kinds of gallstones.
You also used Fourier-transform infrared (FT-IR) spectroscopy in this study. What was the role of FT-IR? How did you accurately interpret or make assignments for the infrared spectra?
Before an elemental study is performed, it is very important to classify stones, because the elemental composition of stones also depends on their types. To classify stones into different categories, we have used FT-IR spectroscopy, which is a very sensitive and powerful analytical tool to study molecular composition of complex samples. In this study, gallstones were categorized into cholesterol and pigmented types based on their relative cholesterol and bilirubin content. The presence of chemical constituents (cholesterol and bilirubin) of stones was confirmed with the presence of vibrational frequencies observed in the fingerprint region. With the help of literature values, accurate absorption peaks were identified and assignments were made in their respective infrared spectra.
What results did you achieve? What did this study show in terms of the feasibility or advantages of WDXRF for such analysis?
Using WDXRF, we achieved very accurate and reliable quantitative data on heavy and carcinogenic elements present in gallstones that were collected from patients of different age groups and with different dietary habits. We found that the stones containing higher amounts of phosphorous also contain higher amount of calcium. The presence of other elements and their potential role was also discussed in the present study. Also, the data obtained in the present work using WDXRF is well correlated with other technique like AA and LIBS, which indicates the feasibility of its routine application to analyze stone samples in the field of gastroenterology and urology.
How does this study help us understand the etiopathogenesis (or cause) of gallstones? Are there many possibly pathways in which the body can form these stones?
There are several factors, such as age, sex, obesity, high blood pressure, chronic diarrhea, urinary tract infections, metabolic abnormalities, diabetes and liver disease, intestinal hypomotality and many more, that influence the formation of stones inside various organs of the human body. Besides these, nutritional imbalances in trace and essential minerals, including the quality of water intake, are the main cause for stone formation. However, mineral water containing calcium and magnesium is beneficial for calcium oxalate kidney stone patients. A large number of studies have indicated the role of heavy and toxic elements on stone formation, growth, and development. With this study, we were able to measure trace and heavy elements in stones, which further indicated the potential role of these elements in the cause of stone formation.
In our study, elemental concentrations were found in varying concentrations in gallstone samples, including a very rare element, palladium.
These differences and occurrence of palladium may be due to differences in patients’ geographical locations, diets, and exposure to environmental pollution. Addition of heavy and toxic metals were found in the gallstones of patients who chew or smoke tobacco. Also, the data obtained from WDXRF were found to be in good correlation with the results obtained from AA spectroscopy. Therefore, it has been concluded that smoking and chewing tobacco should be avoided, particularly by patients of gallstone disease, to prevent gallbladder cancer. Further, this kind of systematic study would be useful to formulate future strategies for the treatment and prevention of these stones.
In a previous study, you used a combination of FT-IR spectroscopy and XRF spectroscopy for the analysis of kidney stones (2). What conclusions did you make from the analysis results in terms of the concentrations of the elements you analyzed and the specific etiopathogenesis of the stones?
To analyze kidney stones, we used a combination of FT-IR and WDXRF spectroscopy. To analyze the molecular constituents of kidney stones and their classification, we used FT-IR spectroscopy. The advantage of using WDXRF is its nondestructive capability. Also, the elements could be quantified using the WDXRF standardless software. Using FT-IR spectroscopy, kidney stones were categorized as oxalate or struvite types, based on their major chemical constituents, calcium oxalate and magnesium ammonium phosphate. Using WDXRF, numerous elements including heavy and toxic elements were quantified in oxalate- and struvite-type kidney stones. Our results revealed that the presence and relative concentrations of trace and heavy elements vary, depending on the stone type. The presence of heavy and toxic metals in kidney stones was found to be related to patients’ consumption of meat products, chewing tobacco, herbal supplements, and nephrotoxin products, such as aspirin, non-steroidal anti-inflammatory drugs, and antibiotics. In conclusion, the differences in the elemental concentrations of different stones may be correlated with geographical, environmental, and dietary differences.
How will this work help in understanding the causes of kidney stone development, or in guiding the treatment or diet or other means to prevent the formation of these stones?
Based on our experimental results, it appears that dietary habits are one of the main causes of kidney stone formation and their development. Oxalate stones are rich is oxalate and calcium, whereas struvite stones are rich in magnesium and phosphorous. Thus, after getting signs of stone formation inside the kidneys, patients should take precautions. Patients of hypercalciuria should avoid calcium supplementation and oxalate-rich foods, such as spinach, cauliflower, milk products, chocolate, and tomatoes. Salt intake should also be reduced, because high sodium intake promotes crystallization of calcium salts by reducing concentrations of citrate. According to our study, positive correlations have been found between the occurrence of heavy and toxic elements in the stones of patients who chew tobacco or consume nephrotoxins and herbal supplements. Thus, intake of these products, including alcoholic beverages, should be reduced or avoided by kidney disease patients. Because of the inhibition action of potassium and magnesium, potassium-magnesium citrate also raises urinary citrate, and diminishes stone formation and recurrence.
Also, one should consult a nephrologist for further necessary steps based on one’s medical history after getting symptoms of stone formation, such as urinary blockage, urinary tract infections, or blood in the urine. Despite many medical advances, the role of many heavy and toxic elements are still unclear and poorly understood in nephrology and urology. More research is still needed to explore the role of heavy elements in kidney stone formation.
In an earlier study, you analyzed heterogenous gallstones using both LIBS and WDXRF (3). What new insights did that study reveal?
Because of the unique and complementary capabilities of LIBS and WDXRF, we have used both these techniques to study the heterogeneous nature of cholesterol- and pigment-type gallstones. Pigment gallstones have been found to be richer in heavy and toxic elements than cholesterol stones, which indicated the difference in formation mechanism of the two types of stones. Using WDXRF, we have detected ruthenium in some gallstones, which is very rare in gallstones. This may be due to the geographical region of the patients involved. These were the preliminary reports on the joint use of LIBS and WDXRF, and more studies are being performed on large number of stone samples. Further studies on gallstones and kidneys stones using LIBS and WDXRF are in progress by our group, in order to obtain further information to provide treatment insights and comfort for patients. I would like to thank Dr. Pradeep K. Rai, a senior nephrologist at the Opal Hospital, in Varanasi, India, for his expertise and help in current research activity.
References
(1) B.B.S.Jaswal, P.K. Rai, T. Singh, V. Zorba, and V.K. Singh, X-Ray Spectrom. 48(3), 178–187 (2019). DOI: 10.1002/xrs.3010
(2) V.K. Singh,B.B.S. Jaswal, J.Sharmaa, and P.K. Raib, X-Ray Spectrom. 46(4), 283-291 (2017).
(3). B.B.S. Jaswal, V. Kumar, J. Sharma, P.K. Rai, M.A. Gondal, B. Gondal, and V.K. Singh, Lasers Med. Sci.31(3), 573–579 (2016).

Vivek K. Singh is an assistant professor of physics at Shri Mata Vaishno Devi University in Katra, India. In the last decade, Singh has worked extensively on applications of XRF, LIBS, TOF-SIMS, and FT-IR spectroscopy for the study of biological specimens. His current research interests include multitechnique spectroscopy studies of biological, organic, medicinal plant, agricultural, and food samples, as well as trace and heavy metal determination in biological, food, and medicinal plant samples. He has published more than 70 research papers and review articles and several book chapters. Singh is also the recipient of the prestigious Raman Fellowship awarded by the University Grants Commission of the Government of India for post-doctoral research at Lawrence Berkeley National Laboratory in Berkeley, California, in 2015–2016. Direct correspondence about this interview to spectroscopyedit@mmhgroup.com

Using Spectroscopy to Understand Hawaii’s Hidden Geology
May 8th 2025Researchers from the University of Nevada, Reno, have conducted the most comprehensive subsurface analysis of Hawaiian shield basalts to date, using advanced spectroscopic and geochemical techniques to reveal short-lived hydrothermal alteration processes and establish a new foundation for future volcanic and geothermal studies.
AI Shakes Up Spectroscopy as New Tools Reveal the Secret Life of Molecules
April 14th 2025A leading-edge review led by researchers at Oak Ridge National Laboratory and MIT explores how artificial intelligence is revolutionizing the study of molecular vibrations and phonon dynamics. From infrared and Raman spectroscopy to neutron and X-ray scattering, AI is transforming how scientists interpret vibrational spectra and predict material behaviors.
Advancing Corrosion Resistance in Additively Manufactured Titanium Alloys Through Heat Treatment
April 7th 2025Researchers have demonstrated that heat treatment significantly enhances the corrosion resistance of additively manufactured TC4 titanium alloy by transforming its microstructure, offering valuable insights for aerospace applications.

.png&w=3840&q=75)

.png&w=3840&q=75)



.png&w=3840&q=75)



.png&w=3840&q=75)