Determining Nitrophenol Isomers Using Raman Spectroscopy
Nitrophenol is one of the most highly toxic heavy pollutants in the environment. It has a slow degradation rate, and is harmful to humans even at low concentrations. Therefore, it is crucial to detect nitrophenol quickly, easily, and accurately. In this study, the nitrophenol isomers (o-nitrophenol, m-nitrophenol, and p-nitrophenol), in solid and liquid phases with methanol or acetone as the solvent, were investigated using Raman spectroscopy. The characteristic Raman peaks of nitrophenol isomers were observed. In addition, the intensities of the corresponding Raman peaks for nitrophenol isomers in the liquid phase with methanol or acetone as solvent at different concentrations (0.3%, 0.6%, 1.2%, 1.8%, 2.4%, 3.0%, and 3.6%) were analyzed. It revealed that the intensities increased when the concentrations increased, and the four corresponding linear correlation coefficients between intensities and concentrations were greater than 0.99. Furthermore, the fitted linear relationships were verified by nitrophenol isomers in the liquid phase with methanol or acetone as the solvent at known concentrations (0.9%, 1.5%, 2.1%, 2.7%, and 3.3%), and the average deviations between the measured data and the known values were 4.94%, 2.69%, 4.37%, and 1.86%, respectively.
Nitrophenol isomers (o-nitrophenol, m-nitrophenol, p-nitrophenol) are widely used as analytical reagents or intermediates for a variety of chemicals (for example, in pesticides, medicines, and dyes). Hence, a large amount of nitrophenol isomers in wastewater and gas is discharged into rivers and the atmosphere. Because nitrophenol isomers are highly toxic and have a slow degradation rate (1,2), they are listed as priority control pollutants by the United States Environmental Protection Agency (EPA), and their concentrations in natural water bodies should be controlled (3). Therefore, developing simple and reliable determination methods for nitrophenol isomers is crucial to protecting people and the environment.
A lot of research has been carried out to investigate nitrophenol isomers. Hofmann and others (4) qualitatively determined p- nitrophenol and 2,4-dinitrophenol in cloud water at low concentrations by high performance liquid chromatography–mass spectrometry (HPLC–MS). Dou and others (5) measured p-nitrophenol and other nitrobenzene compounds by combining dispersive solid-phase extraction (SPE) and HPLC, but these techniques are both complex and expensive. Rahman and others (6) detected p-nitrophenol in a phosphate buffer phase with an electrochemical sensor. Li and others (7) measured o- and p-nitrophenol in aqueous samples by preparing reduced graphene oxide-cyclodextrin-chitosan (RGO-CD-CS) sensors, but the fabrication requirements of these sensors are very strict. Xu and others (8) developed a fluorescence sensor with hybrid magnetic cadmium telluride to detect p-nitrophenol, and a linear relationship between relative fluorescence intensities and concentrations of p-nitrophenol was obtained. However, the measurement accuracy of the sensor was influenced by fluorescence quenching. Manuvesh and others (9) determined that the wide absorption band of nitrophenol was at 345 nm, and the absorption cross sections of nitrophenol were obtained within the range of 295~400 nm by cavity attenuation spectroscopy, but qualitative analysis of nitrophenol cannot be achieved.
Because each molecule has an unique Raman spectrum, Raman spectroscopy in recent years has been widely applied in the fields of medicine, food safety, and environmental pollution monitoring (10–12) owing to the advantages of its noninvasive, real-time, label-free, and fast response. Chi (13) investigated the molecular vibration modes of 2,4-dinitrophenol in polycrystalline phase by Fourier transform (FT)-Raman spectroscopy. Zhang and others (14) compared the Raman scattering enhancement effects of trinitrophenol by synthesizing different types of nanoparticles. In this study, the nitrophenol isomers in both the solid and liquid phase with methanol or acetone as the solvents were investigated using Raman spectroscopy. In addition, the relationships between the intensities and concentrations of the corresponding Raman peaks for nitrophenol isomers in liquid phase were explored.
Experimental Results and Discussion
Experimental Setup and Sample Preparation
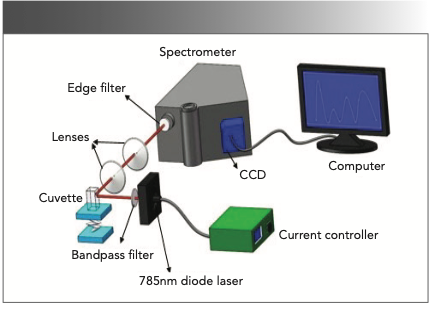
The self-built Raman spectrometer (15) (Figure 1) developed in our previous work was employed here. A diode laser at 785 nm excitation was employed as the light source, and its output power was 50 mW. The spectral resolution of spectrometer (iHR320) was 0.8 cm-1. The detected spectra were recorded with a 1200 g/mm grating. The operating temperature of the charged-coupled detector (CCD) detector (Horiba) was 203 K. To improve the signal-to-noise ratio (S/N) of the spectrometer, the integration time was set at 10 s, with the number of accumulations set to four times. Each sample was measured for five replicate times, and the average spectra were processed for subsequent analysis. The spectrometer was calibrated with anhydrous ethanol before each experiment.
FIGURE 1: Self-built Raman spectrometer, using 785 nm diode laser excitation.
In the work, o-nitrophenol (purity: 99%), m-nitrophenol (purity: 99%), and p-nitrophenol (purity: 99.5%) were purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. Methanol (purity: 99.9%) and acetone (purity: 99.5%) were from Sinopharm Chemical Reagent Co., Ltd. and Shanghai Lingfeng Chemical Reagent Co., Ltd., respectively. Because of the limitation of current experimental conditions in the laboratory, nitrophenol isomers in the gas phase were not investigated in this study. They will be examined in a future study by using near-infrared (IR) laser absorption spectroscopy (16).
System Calibration and Stability
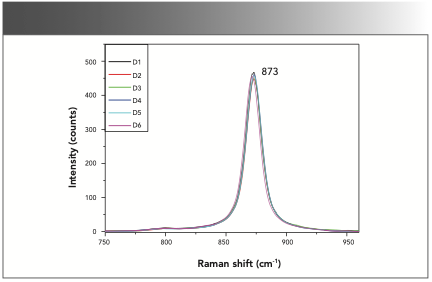
The self-built Raman spectrometer was calibrated daily with anhydrous ethanol (purity: 99.6%). The Raman peak at 873 cm-1 was attributed to the stretching vibration of C-C-O. Figure 2 shows the Raman spectra of anhydrous ethanol for six continuous days (D1–D6). The overlayed spectra of anhydrous ethanol reveals that the spectrometer employed here possessed good stability.
FIGURE 2: The overlayed spectra of anhydrous ethanol detected over six continuous days (D1–D6).
Nitrophenol Isomers in Solid Phase
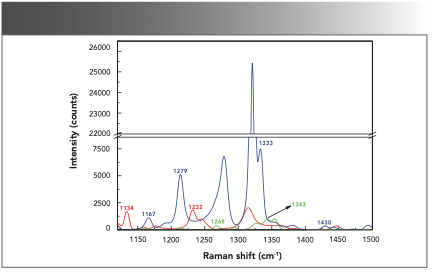
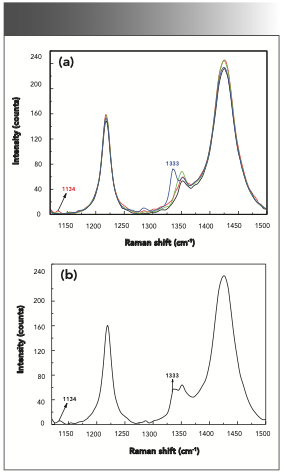
O-, m-, and p-nitrophenol in the solid phase were first detected by the Raman spectrometer. The Raman spectrum of the nitrophenol isomers in the solid phase in the range of 1120~1500 cm-1 are shown in Figure 3. Previous research (14,17) showed that the Raman peaks at 1134, 1167, 1232, 1268, and 1279 cm-1 were attributed to the deformation of C-H. The Raman peaks at 1333 and 1343 cm-1 were caused by the asymmetric stretching vibration of the nitro group, and the Raman peak at 1430 cm-1 was attributed to the symmetric stretching vibration of the nitro group. The study revealed that the Raman peaks at 1134 and 1232 cm-1 were found in the spectrum of o-nitrophenol. The Raman peaks at 1268 and 1343 cm-1 were obtained in the spectrum of m-nitrophenol, and the Raman peaks at 1167, 1279, 1333, and 1430 cm-1 were determined in the spectrum of p-nitrophenol. Based on these characteristic Raman peaks, nitrophenol isomers in the solid phase can be identified.
FIGURE 3: Raman spectra of nitrophenol isomers in the solid phase. O-nitrophenol (red), m-nitrophenol (green), and p-nitrophenol (blue).
Nitrophenol Isomers in Liquid Phase with Methanol as the Solvent
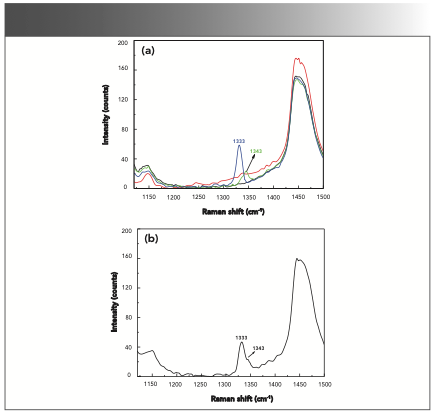
O-, m-, and p-nitrophenol in the liquid phase with methanol as the solvent at a mass fraction of 0.3% were separately investigated. As shown in Figure 4a, a Raman peak at 1343 cm-1 was observed in the Raman spectrum of m-nitrophenol in the liquid phase, and it was consistent with the peak shown in Figure 3. The Raman peak at 1333 cm-1 was also found in the Raman spectrum of p-nitrophenol in the liquid phase, and it also matched well with the Raman peak in the solid phase. Furthermore, the mixture of o-nitrophenol, m-nitrophenol, and p-nitrophenol with methanol at a mass fraction of 0.9% was measured. As shown in Figure 4b, the Raman peaks at 1343 and 1333 cm attributed to m-nitrophenol and p-nitrophenol were also observed and aligned with the results shown in Figure 4a.
FIGURE 4: (a) Raman spectra of methanol (black), methanol and o-nitrophenol (red), methanol and m-nitrophenol (green), and methanol and p-nitrophenol (blue). (b) A mixture of o-, m-, and p-nitrophenol with methanol as the solvent.
Nitrophenol Isomers in Liquid Phase with Acetone as Solvent
O-, m- and p-nitrophenol in the liquid phase with acetone as the solvent at a mass fraction of 0.3% were separately investigated. As shown in Figure 5a, the Raman peak at 1134 cm-1 was observed in the Raman spectrum of o-nitrophenol in the liquid phase, and it was consistent with the Raman peak in the solid phase shown in Figure 3. Raman peak at 1333 cm-1 was observed in the Raman spectrum of p-nitrophenol in the liquid phase, and it matched well with the Raman peak shown in Figure 3. Meanwhile, the mixture of o-nitrophenol, m-nitrophenol, and p-nitrophenol with acetone at a mass fraction of 0.9% was measured. As shown in Figure 5b, the Raman peaks at 1134 cm-1 and 1333 cm-1 attributed to o-nitrophenol and p-nitrophenol were observed, and it matched well with the results shown in Figure 5a.
FIGURE 5: (a) Raman spectra of acetone (black), acetone and o-nitrophenol (red), acetone and m-nitrophenol (green), and acetone and p-nitrophenol (blue). (b) Mixture of o-, m-, and p-nitrophenol with acetone as the solvent.
To summarize the findings, it was found that corresponding Raman peaks can be observed for m-nitrophenol and p-nitrophenol in the liquid phase with methanol as the solvent, and o-nitrophenol and p-nitrophenol in the liquid phase with acetone as the solvent, respectively. These four characteristic Raman peaks (1343 cm-1 of m-nitrophenol with methanol as solvent, 1333 cm-1 of p-nitrophenol with methanol as solvent, 1134 cm-1 of o-nitrophenol with acetone as solvent, and 1333 cm-1 of p-nitrophenol with acetone as solvent at 1333 cm-1) were used for subsequent analysis.
Relationship Between Intensities of Corresponding Raman Peaks and Concentrations for Nitrophenol Isomers in Liquid Phase
The intensity of Raman scattering, IRaman, can be expressed as:
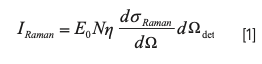
where E0 is the incident light intensity; N is the concentration; η is the optical efficiency of the measurement system; dσRaman/dΩ is the differential scattering cross sections; dΩdet is the receiving solid angle; and V is the volume. Here, when the external environment remains stable and the volume is fixed, the intensity of Raman scattering is linearly dependent on the concentration.
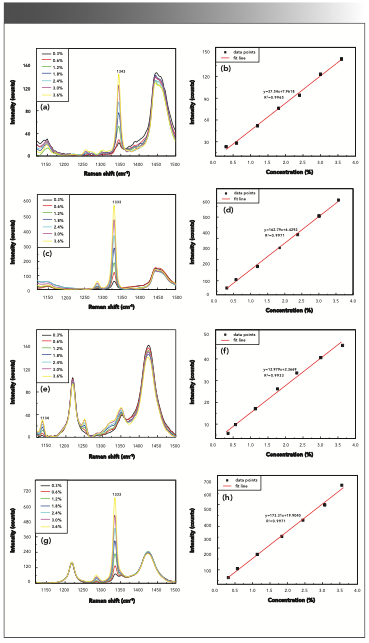
M- and p-nitrophenol in liquid phase with methanol as the solvent at different concentrations (0.3%, 0.6%, 1.2%, 1.8%, 2.4%, 3.0%, and 3.6%), and o- and p-nitrophenol in the liquid phase with acetone as the solvent at different concentrations (0.3%, 0.6%, 1.2%, 1.8%, 2.4%, 3.0%, and 3.6%) were measured. As shown in Figure 6a–h, the intensities of corresponding Raman peaks (1343, 1333, 1134, and 1333 cm-1) increased along with the increasing concentrations. The linear relationships between the intensities and concentrations were found, the corresponding linear correlation coefficients were 0.9965, 0.9971, 0.9933, and 0.9971, respectively. The detection limits of concentrations for m-nitrophenol and p-nitrophenol in the liquid phase with methanol as the solvent, and o-nitrophenol and p-nitro- phenol in the liquid phase with acetone as the solvent were 0.10%, 0.03%, 0.07%, and 0.20%, respectively.
FIGURE 6: (a) Raman spectra of m-nitrophenol in the liquid phase with methanol as the solvent, and (b) linear fit between Raman intensities and concentrations. (c) Raman spectra of p-nitrophenol in the liquid phase with methanol as the solvent, and (d) linear fit between Raman intensities and concentrations. (e) Raman spectra of o-nitrophenol in liquid phase with acetone as solvent, and (f) linear fit between Raman intensities and concentrations. (g) Raman spectra of p-nitrophenol in liquid phase with acetone as solvent, and (h) linear fit between Raman intensities and concentrations.
Validation
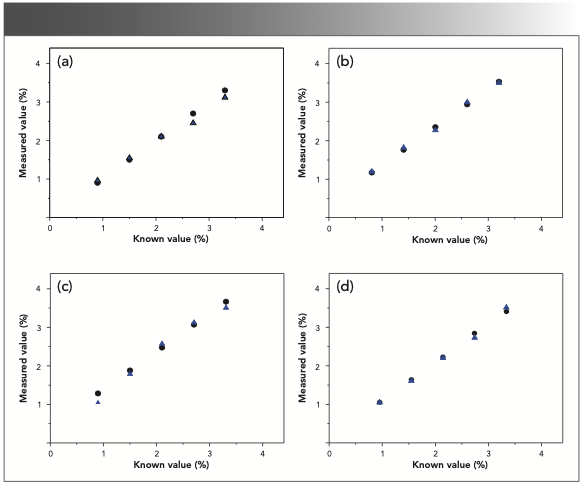
To verify the accuracy of four fitted linear relationships between Raman intensities and concentrations, nitrophenol isomers in the liquid phase with methanol or acetone as the solvent at known concentrations (0.9%, 1.5%, 2.1%, 2.7%, and 3.3%) were investigated, and the measured concentrations calculated by the fitted linear relationships were compared with the known concentrations. As observed in Figure 7a–d, the circle symbol represents the known values of concentrations, and the triangle symbol represents the measured values of concentrations. It revealed that these measured values of concentration were in good agreement with the known values, and average deviations were 4.94%, 2.69%, 4.37%, and 1.86%, respectively.
FIGURE 7: Comparison of known values and measured data of concentrations of nitrophenol isomers in liquid phase. (a) m-nitrophenol with methanol as solvent; (b) p-nitrophenol with methanol as solvent; (c) o-nitrophenol with acetone as solvent; and (d) p-nitrophenol with acetone as solvent. The blue triangle is measured value (%), and the black square is the known value (%).
Conclusions
In this paper, the nitrophenol isomers in solid phase and in liquid phase with methanol or acetone as solvent were investigated by Raman spectroscopy. It showed that several Raman peaks of nitrophenol isomers, except o-nitrophenol with methanol as the solvent and m-nitrophenol with acetone as the solvent, can be obtained. Meanwhile, m-nitrophenol and p-nitrophenol in the liquid phase with methanol as the solvent, and o-nitrophenol and p-nitrophenolin in liquid phase with acetone as the solvent at different concentrations (0.3%, 0.6%, 1.2%, 1.8%, 2.4%, 3.0%, and 3.6%) were investigated. It revealed that the intensities of four corresponding Raman peaks increased with the increasing concentrations linearly, and the four linear correlation coefficients between intensities and concentrations were greater than 0.99. In addition, the fitted linear relationships were verified by nitrophenol isomers in liquid phase with methanol and acetone as the solvent at known concentrations (0.9%, 1.5%, 2.1%, 2.7%, and 3.3%), and it was found that the measured concentrations aligned with the known values, and average deviations between the measured data and the known values were 4.94%, 2.69%, 4.37%, and 1.86%, respectively. This study provides a foundation for determining nitrophenol isomers in environmental monitoring.
Acknowledgment
This work was supported by National Nature Science Foundation of China (NSFC) (Grant Nos. 51676130, 51776129, 51306123) and State Key Laboratory of Applied Optics Open Fund (SKLAO-201909).
References
(1) A. Chatzimarkou, T.G. Chatzimitakos, A. Kasouni, L. Sygellou, A. Avgeropoulos, and C.D. Stalikas, Sens. Actuators B 258, 1152–1160 (2018).
(2) B. Thirumalraj, C. Rajkumar, S.M. Chen, and K.Y. Lin, J. Colloid Interface Sci. 499, 83–92 (2017).
(3) V. Uberoi and S.K Bhattacharya, Water Environ. Res. 69, 146–156 (1997).
(4) D. Hofmann, F. Hartmann, and H. Herrmann, Anal. Biochem. Chem. 391, 161–169 (2008).
(5) Y.N. Dou, L. Guo, G.L. Li, X.X. Lv, L. Xia, and J.M. You, Microchem. J. 146, 366–373 (2019).
(6) M.M. Rahman, H.M. Marwani, F.K. Algethami, A.M. Asiri, S.A. Hameed, and B. Alhogbi, Environ. Nanotechnol. Monit. Manage. 8, 73–82 (2017).
(7) C.K. Li, Z.L. Wu, H. Yang, L. Deng, and X.Q. Chen, Sens. Actuators B 251, 446–454 (2017).
(8) W.Z. Xu, C.X. Qiu, and W.H. Huang, Chin. J. Anal. Chem. 46, 1222–1230 (2018).
(9) S. Manuvesh and L. Zhu, J. Phys. Chem. 120, 9958–9967 (2016).
(10) F. Wei and Z.G. Zou, Chin. J. Electron. Devic. 41, 1136–1140 (2018).
(11) C.J. Strachan, T. Rades, K.C. Gordon, and J. Rantanen, J. Pharm. Pharmacol. 59, 179–192 (2007).
(12) D.V. Petrov, I.I. Matrosov, and A.R. Zaripov, J. Mol. Spectrosc. 348, 137–141 (2018).
(13) V. Chi, Chem. Phys. 300, 1–11 (2004).
(14) Z. Zhang, X.H. Liang, C.Z. He, C. Zhang, R. Xiao, and H. Cheng, Laser Optoelectron. Progress 52, 309–315 (2015).
(15) C. Zhao, R. Li, H.N. Yang, C.X. Shen, X.S. Cai, and C.F. Luo, Chin. J. Lasers 44, 294–300 (2017).
(16) X.Q. Guo, F. Zheng, C.L. Li, X.F. Yang, S.P. Liu, J.L. Wei, X.B. Qiu, and Q.S. He, Opt. Laser Eng. 115, 243–248 (2019).
(17) P. Srinivasan, M. Gunasekaran, T. Kanagasekaran, R. Gopalakrishnan, and P. Ramasamy, J. Cryst. Growth 289, 639–646 (2005).
Wenyou Qiao, Mengmeng Xing, Huinan Yang, Jun Chen, and Mingxu Su are with the School of Energy and Power Engineering at the University of Shanghai for Science and Technology, in Shanghai, China. Jin Tao is with the State Key Laboratory of Applied Optics at the Chinese Academy of Sciences in Changchun, China. Li Peng is with the Shanghai Customs District P.R. China in Shanghai, China. Direct correspondence to Huinan Yang at: yanghuinan@usst.edu.cn

AI-Powered SERS Spectroscopy Breakthrough Boosts Safety of Medicinal Food Products
April 16th 2025A new deep learning-enhanced spectroscopic platform—SERSome—developed by researchers in China and Finland, identifies medicinal and edible homologs (MEHs) with 98% accuracy. This innovation could revolutionize safety and quality control in the growing MEH market.
New Raman Spectroscopy Method Enhances Real-Time Monitoring Across Fermentation Processes
April 15th 2025Researchers at Delft University of Technology have developed a novel method using single compound spectra to enhance the transferability and accuracy of Raman spectroscopy models for real-time fermentation monitoring.
Nanometer-Scale Studies Using Tip Enhanced Raman Spectroscopy
February 8th 2013Volker Deckert, the winner of the 2013 Charles Mann Award, is advancing the use of tip enhanced Raman spectroscopy (TERS) to push the lateral resolution of vibrational spectroscopy well below the Abbe limit, to achieve single-molecule sensitivity. Because the tip can be moved with sub-nanometer precision, structural information with unmatched spatial resolution can be achieved without the need of specific labels.