FT-IR Spectroscopy for Identification of Biological Stains for Forensic Purposes
Special Issues
For analyzing unknown biological stains in a forensic setting, Fourier transform infrared spectroscopy has many advantages. It can provide confirmatory identification of the fluid type, can estimate the stain’s age, discriminate species, and can be used on samples found on various substrates and exposed to various environmental conditions.
Given the large variety of evidence that can be found at a crime scene, a universal method for initial screenings to determine what samples to collect and document for further analysis would be useful. Traces of body fluids are a crucial form of forensic evidence that provides information about potential suspects and victims. It is essential for the screening test to be nondestructive and preserve the evidence for further analysis. Fourier transform infrared (FT-IR) spectroscopy is emerging as the technique of choice for nondestructive analysis of the chemical composition of unknown biological stains. This method is advantageous over most current biochemical tests because of its high specificity, universality, nonconsumptive nature, and potential for quick in situ analysis where the stain is examined directly on the substrate. Here, we provide a brief overview of the novel applications of FT-IR spectroscopy for forensic analysis of body fluid traces. In addition to the confirmatory identification of the fluid type, this technique helps estimate the stain’s age, as well as discriminate species. We also describe the application of FT-IR spectroscopy to biological stains on various substrates and samples exposed to specific environmental conditions to imitate forensically relevant scenarios.
Current forensic practices pay special attention to body fluid traces as the main source of individual DNA evidence. It is extremely important to identify the biological fluids, and develop a potential scenario of how their traces got to the crime scene. Traditionally, forensic investigation of body fluids followed several steps: visual examination, presumptive tests, confirmatory tests, and DNA analysis (1). However, to save time, law enforcement practitioners proceed directly to DNA analysis these days (2,3). DNA typing is relatively expensive and time consuming, which consequently limits the amount of evidence being processed and ultimately contributes to significant backlogs (2).
Very often, it is challenging for investigators to locate biological stains, depending on the color appearance on the substrate where the stain was deposited. To visualize potential body fluids, alternate light sources are often used (4–6). Some alternate light sources can even discover stains that were cleaned or wiped. Presumptive and confirmatory assays are specific to individual body fluids, since they rely on the reaction of chemical components characteristic to a body fluid with a reagent or antibody. For example, hemoglobin is the most targeted component for blood, amylase for saliva, urea or creatinine for urine, acid phosphatase or prostate-specific antigen for semen, and glycogenated epithelial cells for vaginal fluid (2, 4–6). Both presumptive and confirmatory tests can still provide false positive results for other substances containing these or similar characteristic biochemicals. Because these tests require either chemical reaction or antibody–antigen interaction, they destroy the sample, preventing its further use. This sample loss may be an issue if a limited amount of evidence was available at the scene of the crime. Therefore, there is a great need for a universal, nondestructive, rapid, and in situ method for identification and analysis of body fluid traces.
Fourier transform infrared (FT-IR) spectroscopy is a technique already being used during forensic investigations for analysis of physical evidence (such as controlled substances, fibers, and paint) (1). A significant advantage of this technique is its nondestructive nature and specific signature for different sample types based on their chemical composition.
The advantages of FT-IR spectroscopy, as well as other vibrational spectroscopic techniques for various forensic applications, have been recently overviewed by Muro and colleagues (7). The analysis of body fluids using vibrational spectroscopy for a wide spectrum of applications has also been reported (8). Novel methods for forensic body fluid identification and characterization, which include vibrational spectroscopy, have also been reviewed (9,10). Aforementioned review articles cover many topics, and prove that research using FT-IR spectroscopy for body fluid examination is rapidly growing. Here, we briefly overview recent studies on the identification and analysis of body fluid traces using FT-IR spectroscopy for forensic purposes.
Identification of Body Fluid Traces
Since the identification of biological traces can still be problematic for forensic investigators, a nondestructive, universal, and ideally portable method is desired. Elkins (11) was the first to show the potential of FT-IR spectroscopy with attenuated total reflection (ATR) accessory for identification of body fluids and materials. This was done by using unique vibrational signatures of different substances. In this work, spectra of the following biological materials were evaluated: blood, cerumen, feces, fingernails, fingerprints, hair, nasal mucus, vaginal mucus, saliva, semen, and urine. The samples were analyzed in their pure form (as liquid or solid samples) as well as deposited on various substrates imitating forensic samples. The amide I peaks (1700–1600 cm-1) and fingerprint region (<1500 cm-1) were found to be the most informative features for discrimination between different samples.
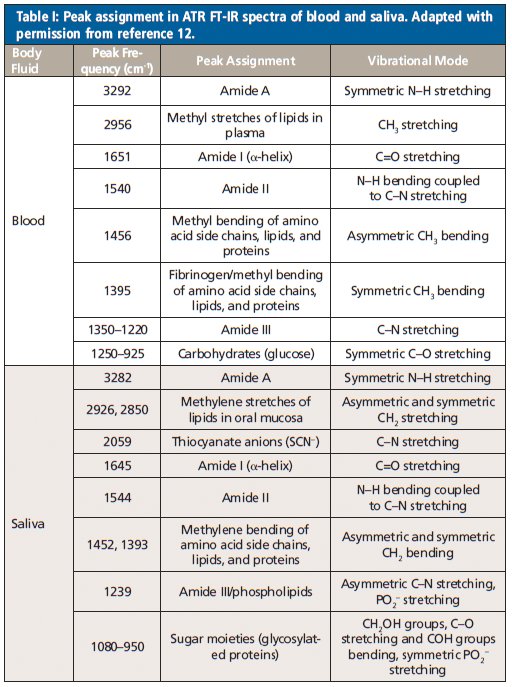
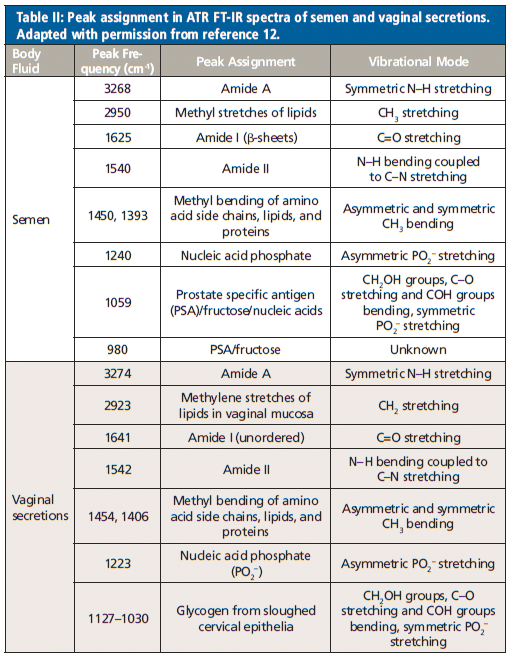
Orphanou and colleagues (12) investigated the possibility of discrimination between IR signatures of different body fluids. In their work, they used blood, saliva, semen, and vaginal secretions to compare as potential fluids in violent and sexual abuse cases. Spectral regions for biological samples were discussed because they all show contributions from biomolecules: lipids (3000–2800 cm-1), proteins (1700–1600 cm-1 and 1560–1500 cm-1), and nucleic acids (1250–1000 cm-1). The body fluids studied showed different IR spectra because of their different biochemical composition. The data was differentiated based on spectral patterns, peak combination, and peak frequencies. The reported characteristic peak assignment for the body fluids can be seen in Tables I and II.
Forensic practice rarely involves pure body fluids. Therefore, the focus of research moved toward the analysis of traces deposited on different surfaces. Gregório and coauthors (13) analyzed body fluids that were potentially involved in sexual offences deposited on superabsorbent pads. Semen, vaginal fluid, and urine, as well as mixtures, were studied. Semen and vaginal fluid showed different IR signatures in the region between 1800 and 1480 cm-1, which corresponds to vibrations of proteins, and therefore is distinguishable from urine. However, since semen and vaginal fluid each contain proteins, it was difficult to discriminate between these body fluids on substrates based on their IR spectra. In their second study, Gregório and colleagues (14) employed statistical analysis to strengthen the identification process for semen. This approach showed potential advantages for the screening of semen deposited on various substrates.
Because of difficulties of discrimination between some samples of similar composition, Zapata and his coauthors (15) also used chemometrics for data obtained from external reflection FT-IR spectroscopy. Stains of body fluids deposited on fabrics and possible false positive substances were studied. Visual differences in spectra were shown, which were also confirmed by principal component analysis (PCA) and soft independent modeling of class analogy (SIMCA) models by correct classification of unknown stains. None of the possible false positives used here (cosmetics or foodstuff) were misclassified as body fluid. Moreover, different colors of cotton did not interfere with results. These results show that chemometrics and FT-IR spectroscopy can be combined to increase discriminating power.
Takamura and colleagues (16) developed a statistical method based on IR spectra of various body fluids (blood, saliva, semen, urine, and sweat). The combination of partial least squares discriminant analysis (PLSDA), linear discriminant analysis (LDA), and Q statistics was employed. The dichotomous classification tree for the ATR FT-IR spectra of five body fluid types, based on hierarchical clustering analysis, can be seen in Figure 1. The method was successfully applied for fresh body fluid samples, and for non-body fluid materials which could potentially interfere with routine crime scene investigation. Samples subjected to an aging process were more difficult to classify. However, the approach showed overall great performance with potential for practical forensic applications.
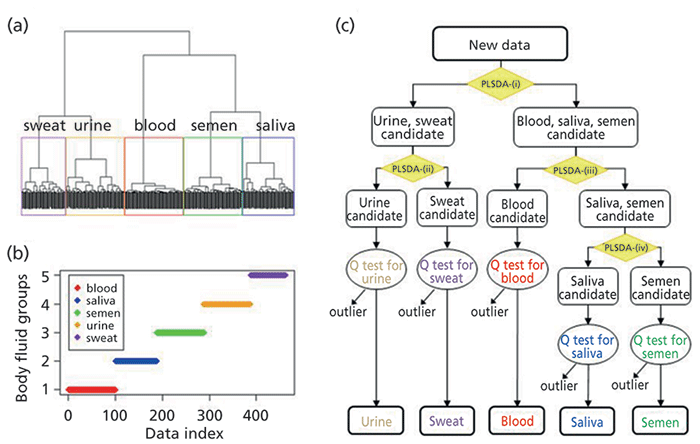
Figure 1: Development of a dichotomous classification tree for the ATR FT-IR spectra of five body fluid (BF) types based on hierarchical clustering analysis: (a) A dendrogram established for the PLSDA scores of 1-day BF spectra predicted by the 1day-5class PLS-LDA-Q model. Borders drawn in the dendrogram indicate clusters of data from each BF group, peripheral blood (red), saliva (blue), semen (green), urine (orange), and sweat (purple). (b) Assignment of the data from each spectrum into the respective BF group. (c) A discrimination scheme using a dichotomous classification tree followed by Q-tests for each type of BF. The dichotomous classification tree was built according to the dendrogram. Four 2-class PLSDA models for the corresponding BF groups were set at each node of the tree: (i) blood-saliva-semen versus urine-sweat using four latent variables (LVs), (ii) urine versus sweat using four LVs, (iii) blood versus saliva-semen using two LVs, and (iv) saliva versus semen using three LVs. Adapted with permission from reference 16.
Studies presented here show great potential of FT-IR spectroscopic techniques to be utilized for body fluid identification with promise for analyzing samples on various substrates. Additionally, the method can be paired with chemometrics to add statistical confidence to findings. It would be extremely beneficial for forensic practical applications to investigate the performance of portable, or even handheld, instruments which are already commercially available. This would also require investigating the subtraction of substrate (like polymers or fibers) to analyze the sample with minimal manipulation by leaving it in place.
Blood Analysis
Since blood is a biological fluid often found at the scene of a violent crime, its analysis was the main focus for many research groups. One of the largest opportunities for adding forensic intelligence is the potential to determine the age of a bloodstain (2). Aging bloodstains can add tremendous value to investigations by targeting valuable biological material from previously deposited material, which will assist in screening and interpretation (2). Edelman and colleagues (17), in addition to visible–near infrared (NIR) spectroscopic analysis of blood, studied the stain's age using Fourier transform NIR spectroscopy. The results showed changes of spectra within the first days of aging. During the first 10 min, when the stain was still fresh, the spectra were dominated by the water contribution. As time progressed, the spectra showed more visible peaks coming from biomolecules.
The sample's age was also studied by Zou and coauthors (18), where the combination of vibrational spectroscopic techniques (that is, ATR FT-IR and Raman spectroscopy) was used. The analysis of blood and semen samples was targeted. The authors were able to select peaks that are characteristic for either blood or semen, and therefore distinguish between these two. For the stain age discrimination, the proposed method was based on the ratio of two specific peaks for each biological fluid: 1531 and 1635 cm-1 for blood, and 1240 and 1633 cm-1 for semen.
Zhang and colleagues (19) examined the changes in blood spectra as the stain dried out. Researchers measured peaks at 3308 cm-1 and 1541 cm-1, and prepared correlation analyses. Human and rat blood samples were subjected for investigation under specific environmental conditions: temperature, humidity, and sample concentration. Logistic regression analysis showed a correlation between absorbance at 3308 cm-1 and the sample's age.
Lin and colleagues (20) brought the estimation of the age of bloodstains to the next level. They analyzed samples applied to indoor and outdoor crime scene conditions. The examined stains ages were between 0.25 and 107 days. For validation purposes, the partial least squares regression (PLSR) models were built to estimate the age of a sample (Figure 2). Furthermore, PLSDA models were built to distinguish between fresh (up to one day old) and older samples. For both indoor and outdoor conditions, the PLSDA models showed accuracy of 0.99 for external validation.
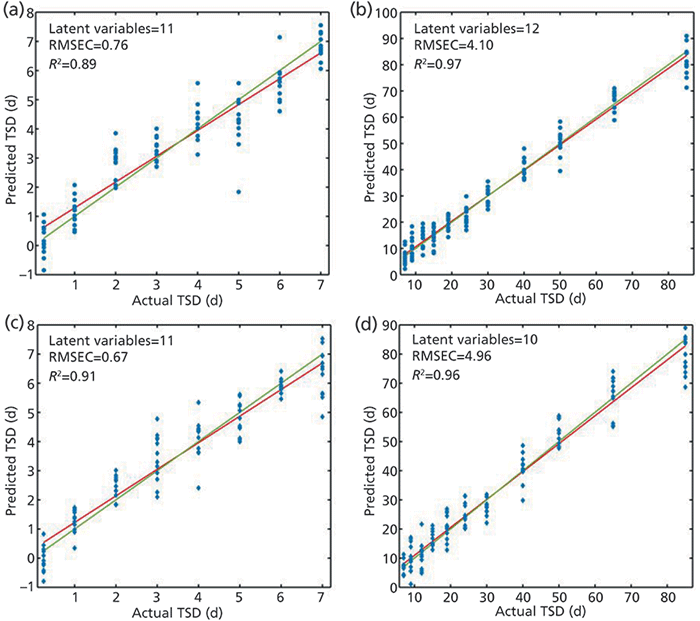
Figure 2: PLSR plots for indoor bloodstain samples in the (a) 0.25–7 d and (b) 7–85 d periods and outdoor bloodstain samples in the (c) 0.25–7 d and (d) 7–85 d periods showing the calibrated age predictions versus the actual age. Adapted with permission from reference 20.
Very frequently, there is a good chance that the size of a sample recovered from a crime scene may be limited, which means establishing the limit of detection of a potential technique is very important. Nonconsumptive in situ testing preserves a sample so maximum biological material is available for subsequent testing such as DNA analysis. Lu and coauthors (21) examined the limit of detection for blood on fabrics using ATR FT-IR spectroscopy. To calibrate spectra of blood dilutions, PLSR was used. The limit of detection for blood on fabrics was found to be 0.0010 µg for cotton, 0.0077 µg for nylon, 0.011 µg for acrylic, and 0.0066 µg for polyester. The same group reported a better limit of detection for blood deposited on the last two (acrylic and polyester) using infrared diffuse reflection spectroscopy (22). However, it was noted that ATR FT-IR spectroscopy provides extreme surface sensitivity, which leads to the improved limit of detection.
Currently, the discrimination between menstrual and peripheral blood on site is challenging for forensic investigators. Menstrual blood is a mixture of peripheral blood and vaginal discharge, and there is no reliable test that can distinguish these two blood types in current forensic practices (2). It would be extremely beneficial for cases involving violence and sexual assault to distinguish those two body fluids. Quinn and Elkins (23) examined the capability of ATR FT-IR spectroscopy for simultaneous discrimination between several body fluids, with emphasis of differentiation between menstrual and peripheral blood. The study confirmed that tested body fluids (menstrual blood, venous blood, semen, saliva, and breastmilk) have their own characteristic IR signatures. When IR spectra of neat samples of peripheral blood, semen, saliva, and breastmilk from two different donors were compared, they showed minimal to small variation in each body fluid. When menstrual blood spectrum was compared to peripheral blood spectrum, it illustrated an enhanced peak at 1039 cm-1 being assigned to phosphoric acid characteristic for menstrual discharge. However, no fluctuations between menstrual blood spectra from more donors were studied here. The testing of samples deposited on various materials showed the fluids could still potentially be detected, but to a lesser degree than pure samples. This is very dependent on the porosity and weave of the substrate.
Another very important aspect of forensic practice is discrimination between postmortem (after death) and antemortem (before death) blood. Takamura and colleagues (24) introduced a novel multivariate spectral processing for discrimination between these two blood classes. The classification model was prepared based on pure samples of blood, spectra of which can be seen in Figure 3. Then blood samples were prepared on various materials (such as polyester, cotton, and denim). After the signal of blood was extracted from the signal of an entire sample (blood and substrate), the blood signal was used for classification, using a model built on pure samples. The discrimination accuracy was improved by using weighted least squares regression. Overall, the method showed high accuracy for discrimination between postmortem and antemortem blood in the presence of substrate interference.
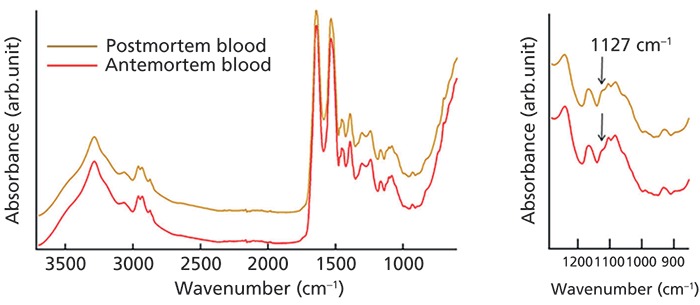
Figure 3: Discrimination of ATR FT-IR of pure postmortem and antemortem blood. Average ATR FT-IR spectra of pure postmortem blood (camel) and antemortem blood (red) (left), and a region representing a distinctive difference between them (right). The 1711–2669 cm-1 region was excluded to avoid interference from the ATR crystal. Adapted with permission from reference 24.
Species Identification
During a forensic investigation, the identification of the bloodstain origin is very important. Current biochemical tests destroy the sample for further analysis, and still have limitations. In the study performed by De Wael and coauthors (25), a combination of spectroscopic techniques was used to analyze blood in particular, so as to discriminate between species. Together with microspectrophotometry and Raman spectroscopy, ATR FT-IR spectroscopy was used to discriminate between human, cat, and dog blood samples. None of these methods proved successful. Spectra of both Raman and ATR FT-IR spectroscopy for the three species presented were not able to be differentiated. This lack of differentiation is not surprising, since all the samples consist of the same components with only quantitative variations within them. Therefore, in the study performed by Mistek and Lednev (26), chemometrics was applied to strengthen classification power. Raw mean IR spectra of different species applied in the study (human, cat, dog, and chicken) can be seen in Figure 4. In their work, PLSDA models were built to discriminate between human and nonhuman blood, and human, cat, and dog. The models allowed for entire separation between classes and external validation of the models performed with unknown samples resulted in 100% prediction accuracy (Figure 5).
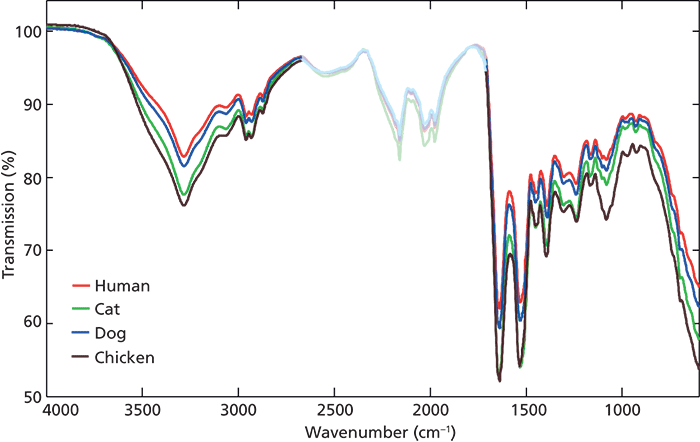
Figure 4: Raw mean IR spectra of human (red line), cat (green line), dog (blue line), and chicken (brown line) blood. The region of 1711–2669 cm-1 was excluded to avoid interference from the diamond ATR crystal. Adapted with permission from reference 26.
Lin and colleagues (27) expanded the study on species identification by adding more animal classes, and performing a study on samples exposed to specific environmental conditions. Samples were subjected to indoor and outdoor conditions, as well as an aging process. Two models were built: one to discriminate between human, other mammals, and domestic fowl, and the other to identify species specifically (human, rat, rabbit, dog, chicken, and duck). Both models illustrated complete separation between classes; no false positive or false negative assignments were observed. For external validation, which was performed using samples exposed to environmental conditions, the first model showed 100% prediction accuracy, while the species specific model showed 94.2%.
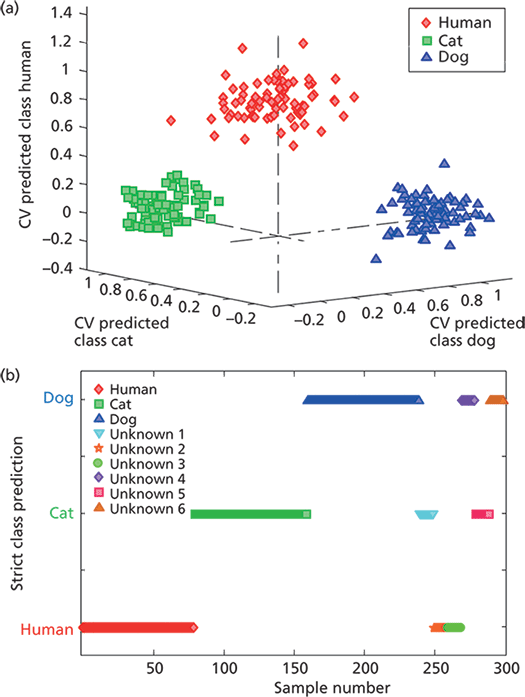
Figure 5: (a) Cross-validated prediction results for the individual species PLSDA model. All three classes are well-separated and grouped together within classes. (b) Strict class prediction results for unknown blood samples (from external experiment) by the species-specific PLSDA model. Each symbol represents a spectrum from human, cat, or dog blood (10 spectra per donor). No unassigned or misclassified spectra are observed. Adapted with permission from reference 26.
It was shown here that FT-IR spectroscopy was applied for a wide spectrum of studies on bloodstains. Researchers used this technique for predicting the age of a bloodstain, estimating the limit of detection for blood, and discriminating between menstrual and peripheral blood, as well as postmortem and antemortem blood. One of the very important aspects in bloodstain analysis is identification of species. The ATR FT-IR spectroscopic approach showed promise for discriminating between human and nonhuman classes, and even between several different animal groups with statistical confidence. It would be extremely beneficial to expand the study with even more species, as it was successfully shown in study where Raman spectroscopy was used (28).
Conclusions
The importance of biological traces that can be found at a crime scene attracts researchers from around the world to apply and examine different approaches for nondestructive identification and analysis of body fluids. This review article was intended to emphasize the great potential of FT-IR spectroscopy for the examination of body fluid traces for forensic purposes.
It was proven that FT-IR spectroscopy is suitable to distinguish between body fluids and various substances which could be mistaken as body fluid. Additionally, this technique shows potential for identification of body fluids that were deposited on substrates. This seems to be most successful by using the combination of FT-IR spectroscopy with statistical data analysis.
Blood is one of the most valuable and frequently found biological fluids on a crime scene. Therefore, its identification and analysis are essential. Many studies focus on the analysis of bloodstains to gather as much information from a sample as possible. A sample's age and limit of detection of blood were investigated using FT-IR spectroscopy. Likewise, discrimination between menstrual and peripheral blood, as well as postmortem and antemortem blood were targeted. ATR FT-IR spectroscopy with chemometrics was successfully applied for discrimination between various species based on blood samples.
Even though FT-IR spectroscopy showed great performance for forensic analysis of body fluids, more investigation needs to be performed before the technique can be used in practical applications. Raman spectroscopy, an alternative vibrational spectroscopic method, showed great potential for human phenotype profiling. The combination of Raman spectroscopy and advanced statistical analysis was applied to identify human sex from bloodstains (29), saliva (30), human race from traces of blood (31), and semen (32), as well as differentiate the age groups of donors (33). It would be important to investigate the capabilities of FT-IR spectroscopy for this application. Our laboratory and others are currently working in this direction. Moreover, portable or handheld instruments should be tested to bring the analysis to a crime scene, saving time and reducing costs of forensic investigation.
This FT-IR spectroscopic technique proved to be advantageous over currently used methods for body fluid analysis. It is particularly beneficial because it is nondestructive and requires a small sample size, and the entire examination can be performed on scene because of the availability of portable instruments. Most importantly, the method is universal and can be used for main body fluids, such as blood, semen, vaginal fluid, saliva, sweat, and urine. FT-IR spectroscopy could have an extremely important role in forensic practice when the method is fully developed.
Acknowledgment
We are grateful to Ray Wickenheiser, Lori Ana Bromberg, and Russell Gettig from the New York State Police Crime Lab System for their advice and valuable discussions.
This project was supported by Awards No. 2017-R2-CX-0006 and NIJ 2017-DN-BX-0135 awarded by the National Institute of Justice, Office of Justice Programs, U.S. Department of Justice. The opinions, findings, and conclusions or recommendations expressed in this publication are those of the authors and do not necessarily reflect those of the U.S. Department of Justice.
References
(1) L. Kobilinsky, Forensic Chemistry Handbook (John Wiley & Sons, Hoboken, New Jersey, 2012).
(2) R. Wickenheiser, L.A. Bromberg, and R. Gettig, New York State Police Crime Laboratory System, Albany, New York, June 2018. Private communication.
(3 National Institute of Justice, 2017, from NIJ.gov: https://www.ncjrs.gov/pdffiles1/nij/250384.pdf.
(4) K. Virkler and I.K. Lednev, Forensic Sci. Int. 188, 1–17 (2009).
(5) J.H. An, K.-J. Shin, W.I. Yang, and H.Y. Lee, BMB Rep.45, 545–553 (2012).
(6) S.A. Harbison and R.I. Fleming, Res. Rep. Forensic Med. Sci.6, 11–23 (2016).
(7) C.K. Muro, K.C. Doty, J. Bueno, L. Halámková, and I.K. Lednev, Anal. Chem. 87, 306–327 (2015).
(8) A.A. Bunaciu, S. FleSchin, V.D. Hoang, and H.Y. Aboul-Enein, Crit. Rev. Anal. Chem. 47, 67–75 (2017).
(9) F. Zapata, M.á. Fernández de la Ossa, and C. García-Ruiz, Trends Anal. Chem. 64, 53–63 (2015).
(10) F. Zapata, I. Gregório, and C. García-Ruiz, J. Forensic Med.1, 101 (2015). Retrieved from https://www.omicsonline.org/open-access/body-fluids-and-spectroscopic-techniques-in-forensics-a-perfect-match-1000101.pdf.
(11) K.M. Elkins, J. Forensic Sci. 56, 1580–1587 (2011).
(12) C.-M. Orphanou, L. Walton-Williams, H. Mountain, and J. Cassella, Forensic Sci. Int. 252, e10–e16 (2015).
(13) I. Gregório, F. Zapata, and C. García-Ruiz, Talanta 162, 634–640 (2017).
(14) I. Gregório, F. Zapata, M. Torre, and C. García-Ruiz, Talanta 174, 853–857 (2017).
(15) F. Zapata, M.á. Fernández de la Ossa, and C. García-Ruiz, Appl. Spectrosc. 70, 654–665 (2016).
(16) A. Takamura, K. Watanabe, T. Akutsu, and T. Ozawa, Sci. Rep. 8, 8459 (2018).
(17) G. Edelman, V. Manti, S.M. van Ruth, T. van Leeuwen, and M. Aalders, Forensic Sci. Int. 220, 239–244 (2012).
(18) Y. Zou, P. Xia, F. Yang, F. Cao, K. Ma, Z. Mi, X. Huang, N. Cai, B. Jiang, X. Zhao, W. Liu, and X. Chen, Anal. Methods 8, 3763–3767 (2016).
(19) Y. Zhang, Q. Wang, B. Li, Z. Wang, C. Li, Y. Yao, P. Huang, and Z. Wang, J. Forensic Sci. 62, 761–767 (2017).
(20) H. Lin, Y. Zhang, Q. Wang, B. Li, P. Huang, and Z. Wang, Sci. Rep. 7, 13254 (2017).
(21) Z. Lu, S.A. DeJong, B.M. Cassidy, R.G. Belliveau, M.L. Myrick, and S.L. Morgan, Appl. Spectrosc. 71, 839–846 (2017).
(22) S.A. DeJong, Z. Lu, B.M. Cassidy, W.L. O’Brien, S.L. Morgan, and M.L. Myrick, Anal. Chem.87, 8740–8747 (2015).
(23) A.A. Quinn and K.M. Elkins, J. Forensic Sci. 62, 197–204 (2017).
(24) A. Takamura, K. Watanabe, T. Akutsu, H. Ikegaya, and T. Ozawa, Anal. Chem. 89, 9797–9804 (2017).
(25) K. De Wael, L. Lepot, F. Gason, and B. Gilbert, Forensic Sci. Int. 180, 37–42 (2008).
(26) E. Mistek and I.K. Lednev, Anal. Bioanal. Chem. 407, 7435–7442 (2015).
(27) H. Lin, Y. Zhang, Q. Wang, B. Li, S. Fan, and Z. Wang, Int. J. Legal Med. 132, 667–674 (2018).
(28) K.C. Doty and I.K. Lednev, Forensic Sci. Int. 282, 204–210 (2018).
(29) A. Sikirzhytskaya, V. Sikirzhytski, and I.K. Lednev, Anal. Chem. 89, 1486–1492 (2017).
(30) C.K. Muro, L. de Souza Fernandes, and I.K. Lednev, Anal. Chem. 88, 12489–12493 (2016).
(31) E. Mistek, L. Halámková, K.C. Doty, C.K. Muro, and I.K. Lednev, Anal. Chem. 88, 7453–7456 (2016).
(32) C.K. Muro and I.K. Lednev, Anal. Chem. 89, 4344–4348 (2017).
(33) K.C. Doty and I.K. Lednev, ACS Cent. Sci. 4, 862-867 (2018).
Ewelina Mistek and Igor K. Lednev are with the Department of Chemistry at the University at Albany, State University of New York, Albany, NY. Direct correspondence to: ilednev@albany.edu

LIBS Illuminates the Hidden Health Risks of Indoor Welding and Soldering
April 23rd 2025A new dual-spectroscopy approach reveals real-time pollution threats in indoor workspaces. Chinese researchers have pioneered the use of laser-induced breakdown spectroscopy (LIBS) and aerosol mass spectrometry to uncover and monitor harmful heavy metal and dust emissions from soldering and welding in real-time. These complementary tools offer a fast, accurate means to evaluate air quality threats in industrial and indoor environments—where people spend most of their time.
NIR Spectroscopy Explored as Sustainable Approach to Detecting Bovine Mastitis
April 23rd 2025A new study published in Applied Food Research demonstrates that near-infrared spectroscopy (NIRS) can effectively detect subclinical bovine mastitis in milk, offering a fast, non-invasive method to guide targeted antibiotic treatment and support sustainable dairy practices.
Smarter Sensors, Cleaner Earth Using AI and IoT for Pollution Monitoring
April 22nd 2025A global research team has detailed how smart sensors, artificial intelligence (AI), machine learning, and Internet of Things (IoT) technologies are transforming the detection and management of environmental pollutants. Their comprehensive review highlights how spectroscopy and sensor networks are now key tools in real-time pollution tracking.