Kinetic Fluorimetric Determination of Formaldehyde by Oxidation of Rhodamine B with Potassium Bromate
Based on the H2SO4-Rhodamine B-KBrO3 system, the kinetic fluorescence method was used to determine formaldehyde. The experimental results show that in the sulfuric acid medium, the fading process of oxidized rhodamine B by potassium bromate is inhibited by the catalytic action of formaldehyde. The results showed that the formaldehyde concentration in the range of 0.4 to 4.4 μmol/L had a good linear relationship with lg (IF/I0F) = 0.01385 c (μmol/L) + 0.02353, the correlation coefficient was 0.9974, and the method detection limit was 0.3027 μmol/L. In the determination of formaldehyde in the synthetical water sample and laundry water, the recoveries were 98.68% and 103.20%. The results were consistent with the certified value.
Formaldehyde can cause great harm to the human body. It can cause skin and mucous membrane irritation. When a high concentration of formaldehyde is inhaled, severe respiratory tract irritation and edema, eye irritation, headaches, and other symptoms can occur. Therefore, it is important to obtain a reliable and accurate formaldehyde result in environmental monitoring. The detection methods of formaldehyde mainly include spectrophotometry (1), kinetic spectrophotometry (2), fluorescence photometry (3), gas chromatography (GC) (4), liquid chromatography (LC) (5,6), and the electrochemical analysis method (7). Among them, kinetic spectrophotometry offers several key advantages as a technique, including high sensitivity, high selectivity, simple operation, and a large linear range. Compared with kinetic spectrophotometry, other methods have their own shortcomings (8–16). Rhodamine B is a red-purple or green crystalline powder. It is easily soluble in alcohol, water, and most organic solvents. In polar solvents, such as ethanol or water, the strong polarity of the solvent changes the molecular structure of rhodamine B, making it a zwitterion R+-. When the pH is less than 3, R+- can be protonated to generate RH+, RH22+, and RH33+. These three cations can form various ion associations with halogen ions, such as Cl- and Br-, such as RHCl, RH2Cl+, and RH2Cl2. On the basis of the existing literature, (17,18), we found that in the sulfuric acid medium, the fading process of rhodamine B oxidation by potassium bromate can be inhibited by the catalysis of formaldehyde. The concentration of formaldehyde has a good linear relationship with lg (IF/I0F) in the range of 0.4–4.4 μmol/L. Based on this, a new method for determining the content of formaldehyde has been established.
Experimental
Apparatus
A F-7100 fluorescence spectrophotometer (Hitachi Ltd., Japan), a BS 110S electronic balance (Beijing Sartorius Instrument System Co., Ltd., China), a U3900H UV spectrophotometer (Hitachi Ltd., Japan), a HH-2 constant temperature water bath (Guohua Electric Co., Ltd.), and a KQ3200E ultrasonic cleaner (Kunshan Ultrasonic Instrument Co., Ltd.) was used in this study.
Reagents
Potassium bromate was acquired from the Tianjin Quartz Clock Factory Baxian Chemical Plant; rhodamine B was acquired from the Tianjin Tianxin Fine Chemical Plant Development Center; the sulfuric acid originated from the Beijing Chemical Plant; formaldehyde came from the Tianjin Fuchen Chemical Reagent Plant; hydrochloric acid came from the Beijing Chemical Plant; nitric acid was acquired from the Baiyin Liangyou Chemical Reagent Co., Ltd.; and acetic acid came from the Tianjin Oboke Chemical Co., Ltd. All reagents were of analytical grade.
A 1.00 × 10-3 mol/L rhodamine B solution, a 0.10 mol/L sulfuric acid solution, and a 0.10 mol/L potassium bromate solution was prepared with the above reagents and pure water.
The formaldehyde standard solution (1 mmol/L) was prepared with a 1.00 mmol/L formaldehyde standard solution with a 37% formaldehyde solution before being calibrated by an iodometric method and diluted with water to a 0.01 mmol/L standard working solution before use.
Experimental Method
This experiment utilized four 25 mL colorimetric tubes (numbered 1–4). In each tube, the 2.0 mL 0.10 mol/L sulfuric acid solution and the 0.50 mL 1.00 × 10-3 mol/L rhodamine B solution were added, respectively. Then, 5.0 mL of the 0.10 mol/L potassium bromate solution were added respectively to the colorimetric tubes numbered 2–4. Finally, the 2.00 mL 1.00 × 10-5 mol/L formaldehyde standard solution was added to the tube labeled 3. The 4.000 mL 1.00 × 10-5 mol/L formaldehyde standard solution was added to the tube labeled number 4, and the volume constant was made. All the colorimetric tubes were placed in a water bath at 90 °C for 15 min. Then, they were removed from the bath and cooled with flowing cold water for 3 min. Finally, the fluorescence intensity IF of the catalytic solution and the fluorescence intensity I0F of the non-catalytic solution at λex = 572 nm and λem = 584 nm were measured, respectively.
Results and Discussion
Excitation Emission Matrix Spectra
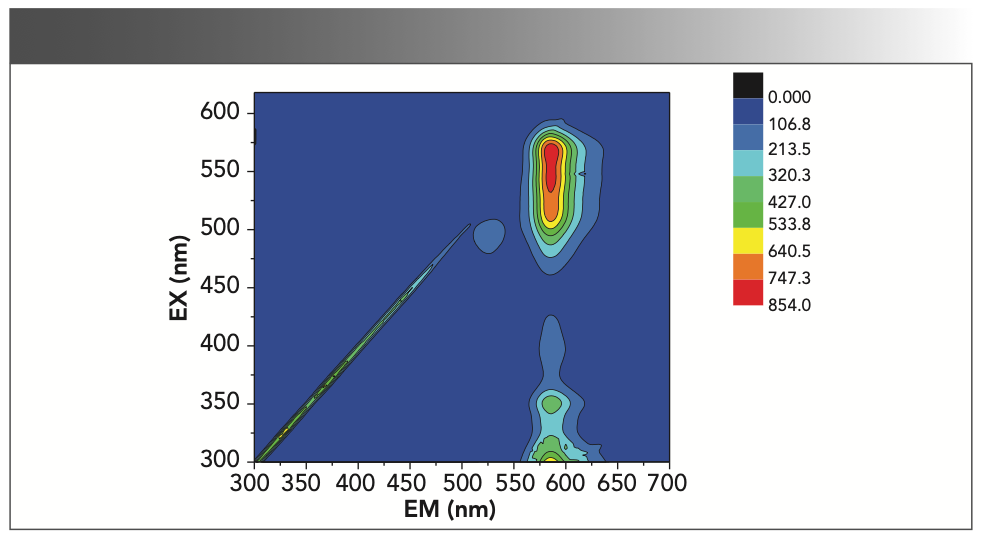
The excitation emission matrix (EEM) spectra of the blank solution without formaldehyde is shown in Figure 1. It can be seen from Figure 1 that when the excitation wavelength is 572 nm and the emission wavelength is 584 nm, the sample has maximum fluorescence intensity.
FIGURE 1: Excitation emission matrix spectra.
Fluorescence Spectra
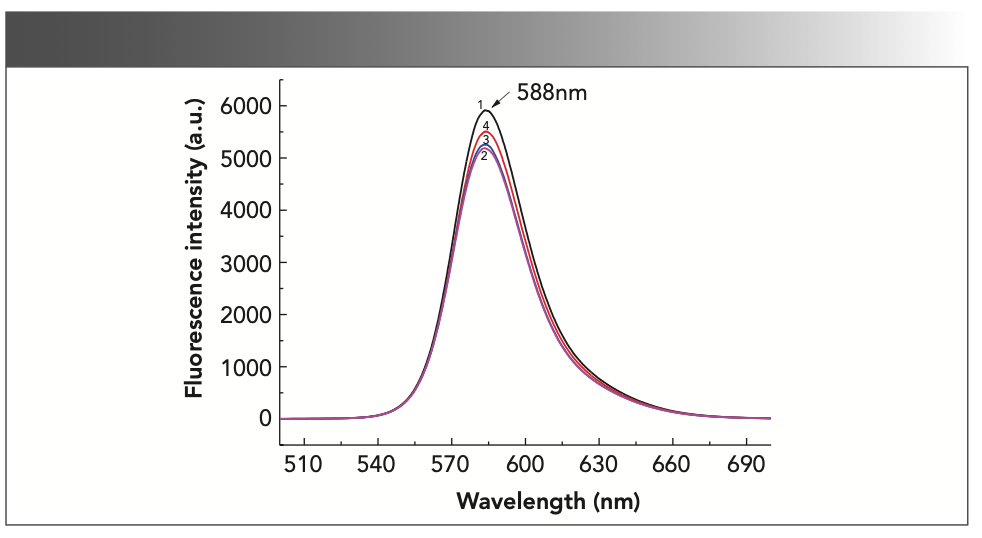
In Figure 2, curves 1 and 2 show that under acidic conditions, the fluorescence intensity of rhodamine B under the action of potassium bromate is significantly reduced, indicating that KBrO3 can oxidize rhodamine B in an acidic medium, with large peak intensity changes. From curve 3 and 4, it can be seen that the fluorescence intensity in the system increases as the amount of formaldehyde increases, indicating that formaldehyde can significantly inhibit the fading reaction, which can be used as a basis for measuring formaldehyde.
FIGURE 2: Fluorescence emission spectra of the rhodamine B system. 1: 2 mL 0.10 mol/L sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B; 2: 1 + 5 mL 0.10 mol/L potassium bromate solution; 3: 2 + 2.00 mL 1.00 × 10-5 mol/L formaldehyde standard solution; 4: 2 + 4.00 mL 1.00 × 10-5 mol/L formaldehyde standard solution.
Optimum Condition for the Reaction
Effect of the Acid Type
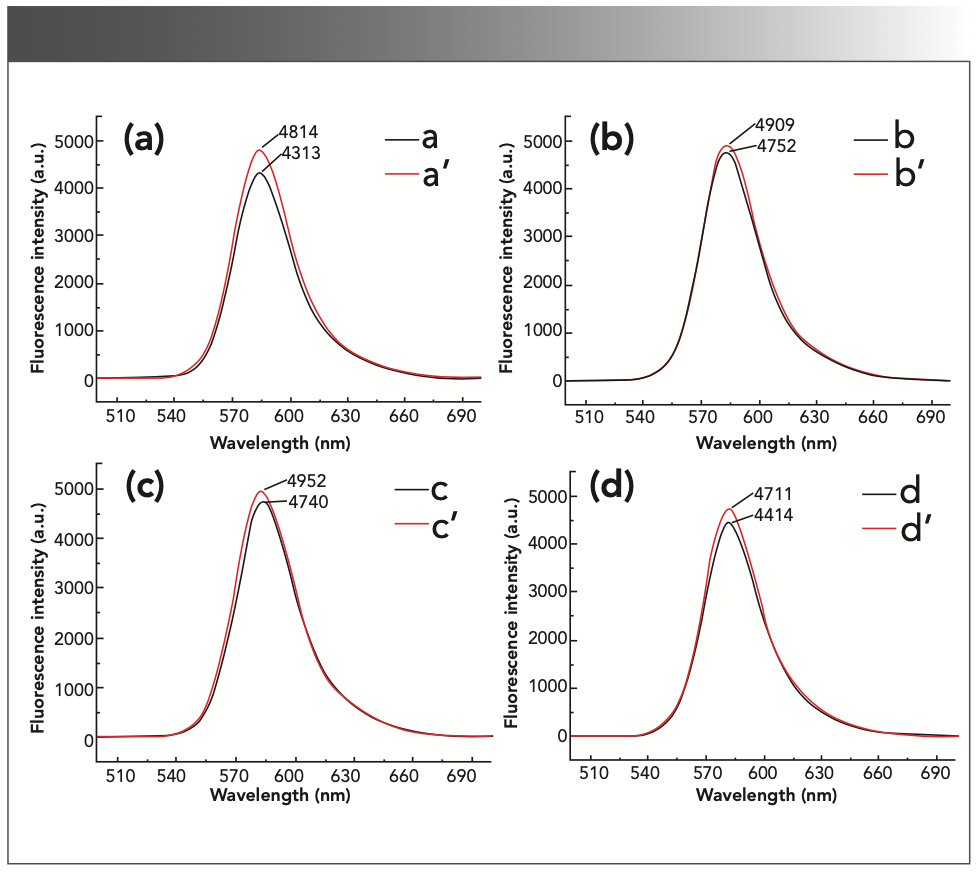
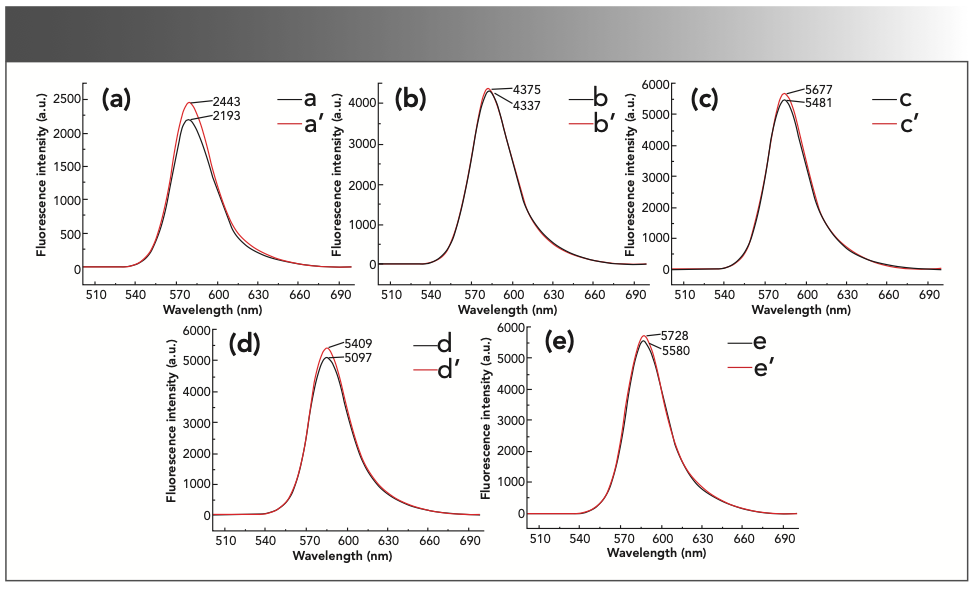
Different types of acids have a greater impact on the experimental results. The experiment investigated the effects of sulfuric acid, hydrochloric acid, nitric acid, and acetic acid on the fluorescence intensity of the reaction product. The results are shown in Figure 3. In the H2SO4 medium, the inhibitory effect of formaldehyde was obvious, and the ΔIF was the largest. In the HNO3, HCl, and acetic acid (AcOH) mediums, the fluorescence intensity of the inhibitory reaction and the non-inhibitory reaction did not change much. Therefore, H2SO4 was used as the reaction medium in the subsequent experiments.
FIGURE 3: The fluorescence intensity of the reaction system in different acid mediums. (a) a’: 2.0 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution; a: a’ + 2.00 mL formaldehyde standard solution. (b) b’: 2.0 mL hydrochloric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution; b: b’ + 2.00 mL formaldehyde standard solution. (c) c’: 2.0 mL nitric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution; c: c’ + 2.00 mL formaldehyde standard solution. (d) d’: 2.0 mL acetic acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution. d: d’ + 2.00 mL formaldehyde standard solution.
Effect of the Sulfuric Acid Concentration
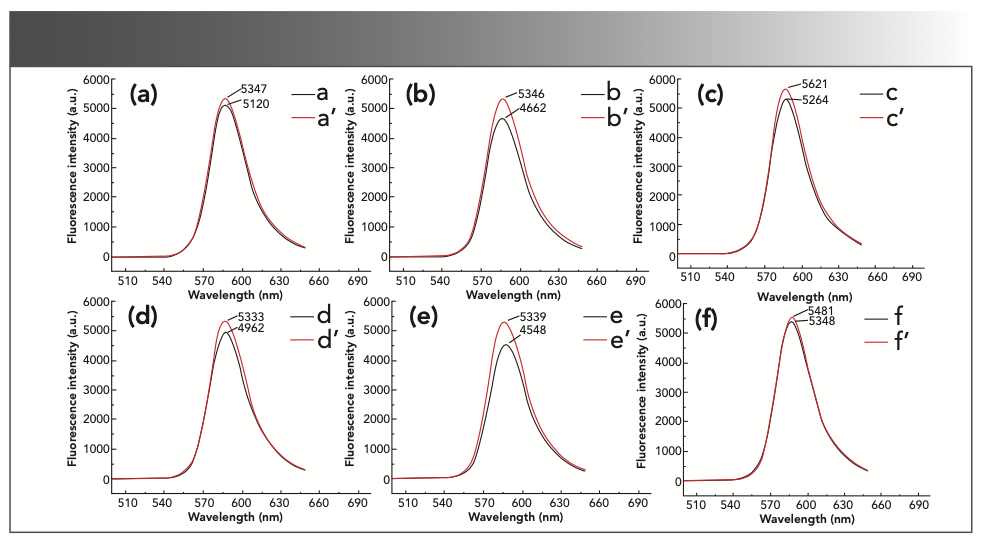
The experiment investigated the effect of the different amounts of sulfuric acid on the fluorescence intensity of the reaction product. The results are shown in Figure 4. When the volume of H2SO4 was 2.50 mL, the inhibitory effect of formaldehyde was obvious, and the ΔIF was the largest. Therefore, the optimum volume of the H2SO4 solution in the subsequent experiments was 2.50 mL.
FIGURE 4: The fluorescence intensity of the reaction system in different volumes of sulfuric acid. (a) a’: 0.5 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution; a: a’ + 2.00 mL formaldehyde standard solution. (b) b’: 1.0 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution; b: b’ + 2.00 mL formaldehyde standard solution. (c) c’: 1.5 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution; c: c’ + 2.00 mL formaldehyde standard solution. (d) d’: 2.0 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution; d: d’ + 2.00 mL formaldehyde standard solution. (e) e’: 2.5 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution; e: e’ + 2.00 mL formaldehyde standard solution. (f) f’: 3.0 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 5 mL 0.10 mol/L potassium bromate solution; f: f’ + 2.00 mL formaldehyde standard solution.
Effect of Potassium Bromate Dosage
Under the above selected conditions, the experiment investigated the influence of different volumes of the potassium bromate solution on the experimental results. The results are shown in Figure 5. When the volume of KBrO3 was 5.0 mL, the inhibitory effect of formaldehyde was obvious, and the ΔIF was the largest. Therefore, the volume of the KBrO3 solution in the subsequent experiments was 5.0 mL.
FIGURE 5: The fluorescence intensity of the reaction system in different volumes of the potassium bromate solution. (a) a’: 1 mL 0.10 mol/L potassium bromate solution + 2.5 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B; a: a’ + 2.00 mL formaldehyde standard solution. (b) b’: 3 mL 0.10 mol/L potassium bromate solution + 2.5 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B; b: b’ + 2.00 mL formaldehyde standard solution. (c) c’: 5 mL 0.10 mol/L potassium bromate solution + 2.5 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B; c: c’ + 2.00 mL formaldehyde standard solution. (d) d’: 7 mL 0.10 mol/L potassium bromate solution + 2.5 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L rhodamine B; d: d’ + 2.00 mL formaldehyde standard solution. (e) e’: 9 mL 0.10 mol/L potassium bromate solution + 2.5 mL sulfuric acid solution + 0.50 mL 1.00 × 10-3 mol/L Rhodamine B; e: e’ + 2.00 mL formaldehyde standard solution.
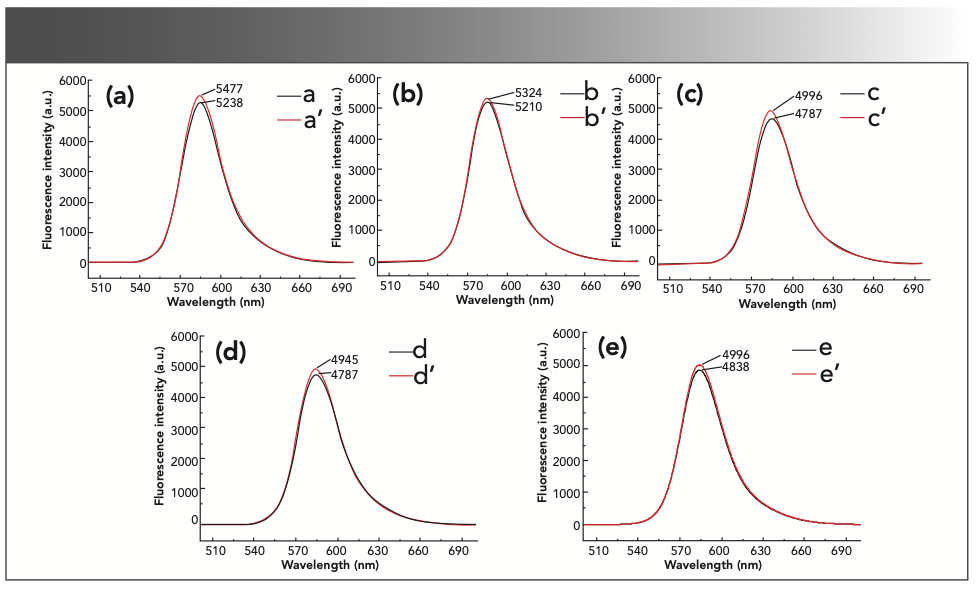
Effect of Rhodamine B Dosage
Under the above selected conditions, the experiment investigated the influence of different volumes of rhodamine B on the experimental results. The results are shown in Figure 6. The fluorescence intensity value of the non-inhibitory reaction gradually increased with the increase of the volume of rhodamine B, but when the volume of rhodamine B was 0.50 mL, the reaction had a higher fluorescence intensity and the inhibitory effect of formaldehyde was obvious, so the volume of the rhodamine B solution in the subsequent experiments was 0.50 mL.
FIGURE 6: The fluorescence intensity of the reaction system in a rhodamine B solution with different volumes. (a) a’: 0.10 mL 1.00 × 10-3 mol/L rhodamine B + 2.5 mL sulfuric acid solution + 5 mL 0.10 mol/L potassium bromate solution; a: a’ + 2.00 mL formaldehyde standard solution. (b) b’: 0.30 mL 1.00 × 10-3 mol/L rhodamine B + 2.5 mL sulfuric acid solution + 5 mL 0.10 mol/L potassium bromate solution; b: b’ + 2.00 mL formaldehyde standard solution. (c) c’: 0.50 mL 1.00 × 10-3 mol/L rhodamine B + 2.5 mL sulfuric acid solution + 5 mL 0.10 mol/L potassium bromate solution; c: c’ + 2.0 mL formaldehyde standard solution. (d) d’: 0.70 mL 1.00 × 10-3 mol/L rhodamine B + 2.5 mL sulfuric acid solution + 5 mL 0.10 mol/L potassium bromate solution; d: d’ + 2.00 mL formaldehyde standard solution. (e) e’: 0.90 mL 1.00 × 10-3 mol/L rhodamine B + 2.5 mL sulfuric acid solution + 5 mL 0.10 mol/L potassium bromate solution; e: e’ + 2.00 mL formaldehyde standard solution.
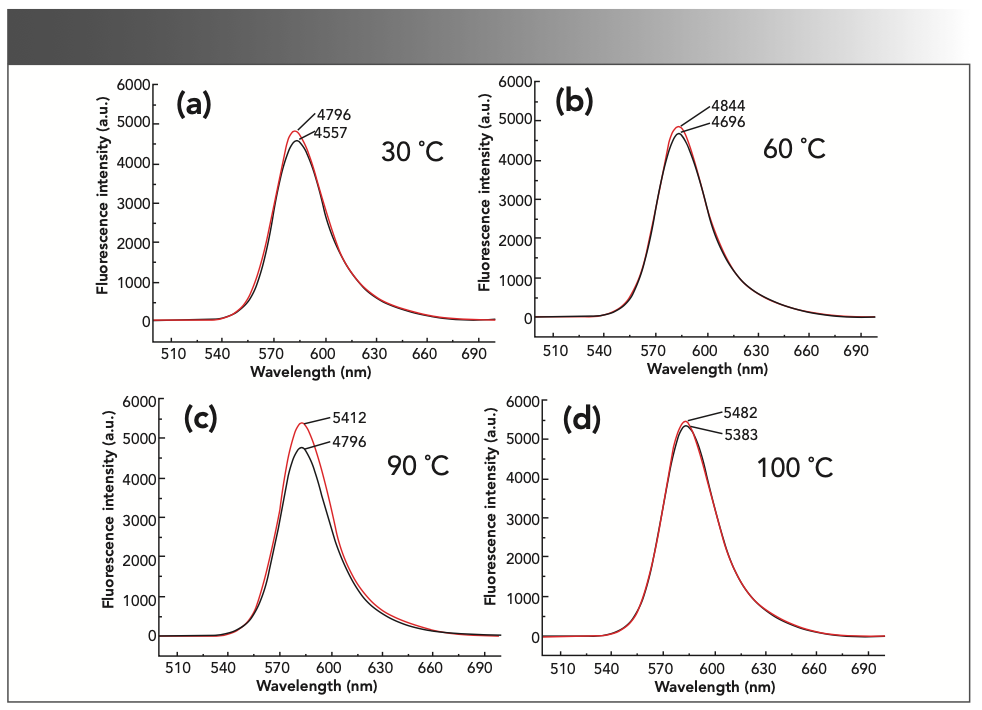
Optimal Reaction Temperature and Time
Under the above selected conditions, the experiment investigated the influence of the water bath temperature on the experimental results. The results are shown in Figure 7. When the temperature was 90 °C, ΔIF had a maximum value, so in the subsequent experiments, the reaction temperature was selected to be heated in a water bath at 90 °C.
FIGURE 7: The fluorescence intensity of the reaction system at different water bath temperatures: (a) 30 °C, (b) 60 °C, (c) 90 °C, and (d) 100 °C.
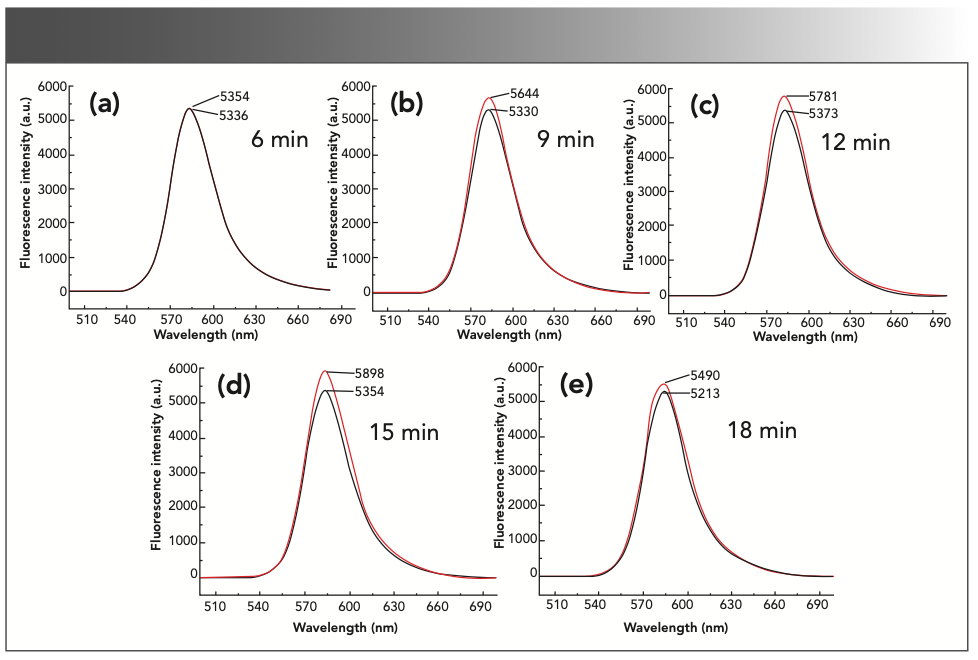
Under the above selected conditions, the experiment investigated the influence of heating temperature on the experimental results. The results are shown in Figure 8. As the reaction time increased, ΔIF first increased and then decreased. When the reaction time was 15 min, ΔIF was relatively stable and had a maximum value, so the reaction time was 15 min in the subsequent experiments.
FIGURE 8: The fluorescence intensity of the reaction system under different reaction times: (a) 6 min; (b) 9 min; (c) 12 min; (d) 15 min; and (e) 18 min.
As a result, the optimum conditions were as follows: 2.50 mL of the dilute H2SO4 solution was added, 5.0 mL of the KBrO3 solution was added, and 0.50 mL of the rhodamine B solution was placed in a 25-mL colorimetric tube and put in a water bath at 90 °C for 15 min before being cooled with cold water for 3 min.
Working Curve and Detection Limit
Drawing of Working Curve
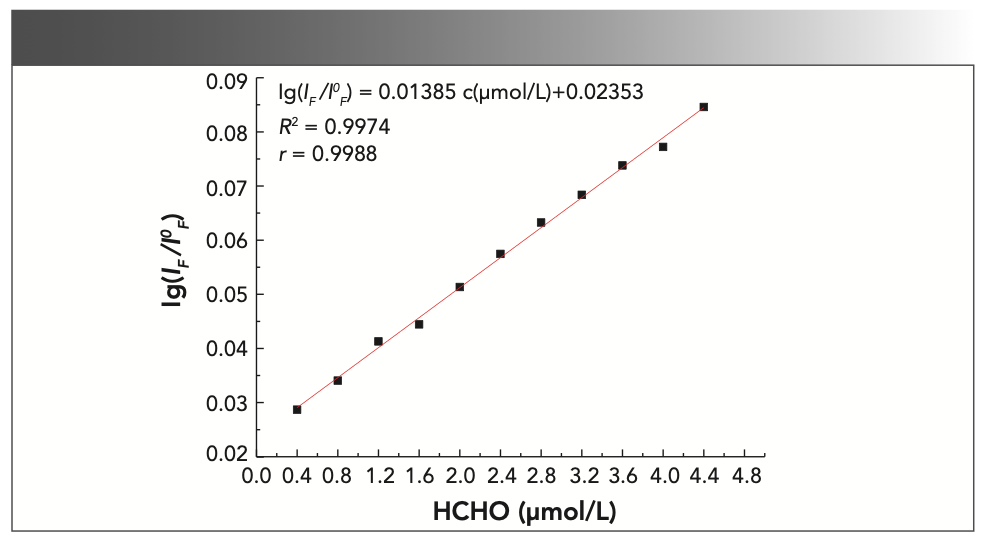
For this part of the experiment, 12 25 mL colorimetric tubes were used and 0, 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, and 11 mL of formaldehyde standard solution was added to them before reacting under the optimal experimental conditions described above. Then, the fluorescence spectrum was measured in the range of 500~700 nm. The experimental results are shown in Figure 9. The results show that when the formaldehyde concentration is 0.4~4.4 μmol/L, the lg (IF/I0F) of the system has a linear relationship with the concentration of formaldehyde, as shown in Figure 10. The linear equation was lg (IF/I0F) = 0.01385 c (μmol/L) + 0.02353, and the maximum linear correlation coefficient was 0.9974.
FIGURE 9: The fluorescence diagram of the standard curve experiment. The image to the right shows a close-up of the peak area.
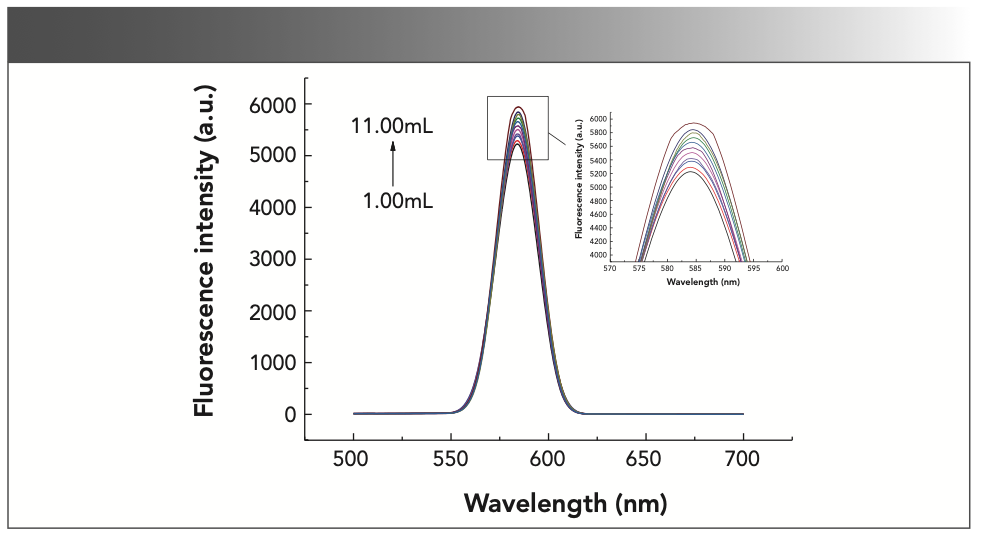
FIGURE 10: The standard working curve with the correlation results.
Determination of Detection Limit
The fluorescence intensity of the 11 blank samples was measured to obtain the standard deviation σ. Then, we divided 3σ by the slope of the standard working curve to obtain a detection limit of 0.3027 μmol/L.
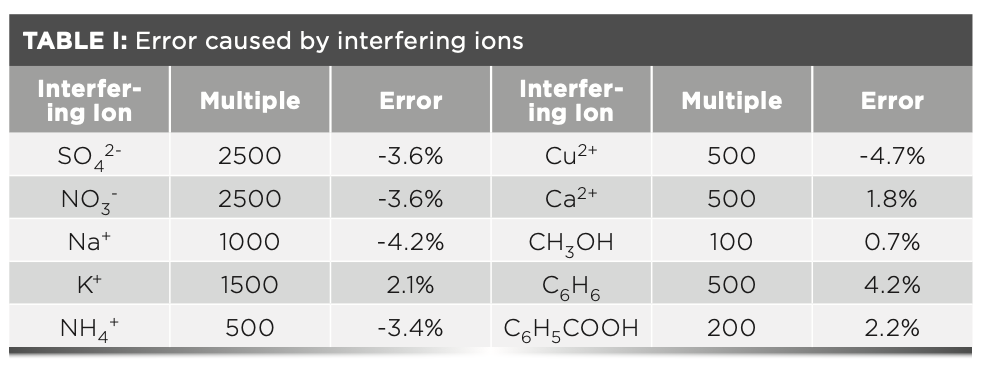
Ion Interference Experiment
The experiment investigated the influence of common ions and compounds on the determination of formaldehyde. The allowable relative error was controlled within ±5%. The allowable multiples of the following interfering ions are shown in Table I.
Possible Reaction Mechanism
In polar solvents such as ethanol or water, the structure of rhodamine B would be changed by the strong polarity of the solvent, making it a zwitterion R+-. When the pH is less than 3, R+- would be protonated to generate RH+. The positively charged RH+ can combine with the negatively charged BrO3- and be oxidized by it. The conjugated structure of rhodamine B was destroyed as the fluorescence intensity of the solution decreases (19–21); its fading process is shown in Figure 11.
FIGURE 11: Schematic illustrating the decreased intensity of rhodamine b.
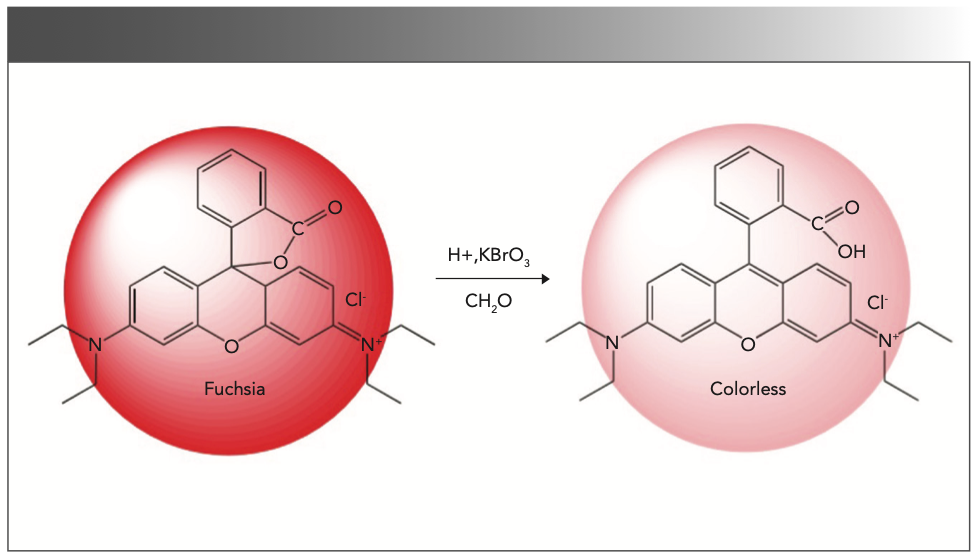
Formaldehyde has weak reducibility and can be oxidized by potassium bromate to formic acid, which can oxidize rhodamine B and generate formaldehyde. Therefore, formaldehyde competes with rhodamine B for the opportunity to combine with potassium bromate, so it can inhibit the fading process of rhodamine B. The fluorescence intensity of the reaction system increases with the increase of the formaldehyde concentration. Within a certain range, lg (IF/I0F) shows a good linear relationship with the concentration of formaldehyde.
Sample Analysis
Analysis of Synthetical Water Sample
A water sample was prepared with the following components: 0.8 μmol/L formaldehyde; 0.1 μmol/L Na2SO4; and 0.2 μmol/L KNO3. Next, an appropriate amount of the laboratory synthetical water sample was taken. Then, 10 mL was measured after filtration and transferred to a 100 mL volumetric flask and diluted with water to the volume and mixed. After that step was completed, 3.0 mL of the sample was added to the 25-mL colorimetric tube before the test was carried out according to the best experimental conditions while the addition experiment was performed at the same time. After the reaction, the fluorescence spectrum was measured in the range of 500~700 nm on a fluorescence spectrophotometer, and the fluorescence intensity value corresponding to 584 nm was taken. Then, the fluorescence intensity value obtained was brought into the standard working curve equation, where the formaldehyde concentration and standard recovery rate was calculated. The results are shown in Table II.
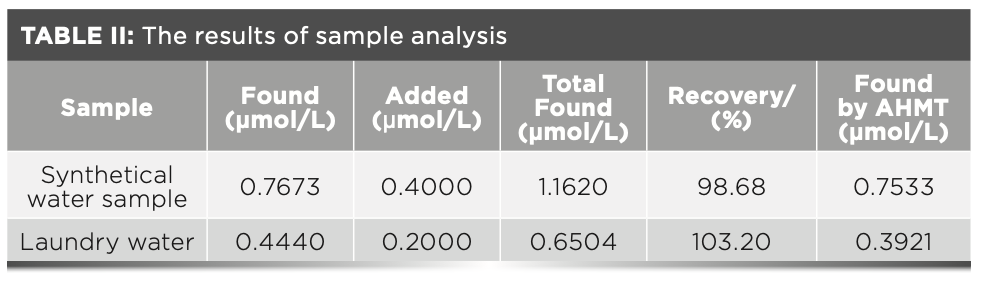
Analysis of Laundry Water Samples
To analyze the laundry water samples, a new piece of clothing was soaked in tap water for 5 h, and 10 mL of washing water was measured with a graduated cylinder. The liquid was then transferred to a 100 mL volumetric flask, and the volume and mix was diluted with water. Then, 3.0 mL of the sample was added to a 25 mL colorimetric tube, and then the procedural steps outlined in “Analysis of Synthetical Water Sample” were repeated. The results are shown in Table II.
Conclusion
In this paper, a kinetic fluorimetric method for the determination of formaldehyde by potassium bromate oxidation of rhodamine B was established. In sulfuric acid medium, the fading process of potassium bromate oxidation of rhodamine B would be inhibited by the catalysis of formaldehyde. As the concentration of formaldehyde increased, the fluorescence intensity gradually increased. In a certain range, lg (IF/I0F) had a good linear relationship with the concentration of formaldehyde, so it could be used as a basis for determining formaldehyde. After defining the optimal experimental conditions, further experiments were carried out on this basis. The experimental results showed that the concentration of formaldehyde had a good linear relationship with lg (IF/I0F) in the range of 0.4 μmol/L to 4.4 μmol/L. The standard working curve was lg (IF/I0F) = 0.01385 c (μmol/L) + 0.02353, the correlation coefficient was 0.9974, and the detection limit of the method was 0.3027 μmol/L. In the determination of formaldehyde in the synthetical water sample and laundry water, the spike-and-recovery rates were 98.68% and 103.20%, respectively, and the results were good.
Acknowledgment
The authors acknowledge the financial support from the National Natural Science Foundation of China (No. 62075007).
References
(1) G.F. Pinto, D.L. Rocha, E.M. Richter, R.A.A. Muñoz, and S.G. da Silva, J. Braz. Chem. Soc. 29(7), 1400–1405 (2018).
(2) A. Afkhami and H. Bagheri, Microchimica Acta. 176, 217–227 (2012).
(3) G. Nájera-Estebanjuan, E.G. Carrillo-Cedillo, and M.P. Cañizares-Macías, Anal. Methods 11(15), 2041–2049 (2019).
(4) X. L. Wang and L. Zhang, IOP Conf. Ser: Earth Environ. 647(1), 012155 (2021). DOI: 10.1088/1755-1315/647/1/012155
(5) A.S. Sebaei, A.M. Gomaa, A.A. El-Zwahry, E.A. Emara, and C.L. Wilkins, Int. J. Anal. Chem. 2018, 2757941 (2018).
(6) B.K. Ma, F.J. Xu, M. He, Y.Q. Lin, et al, Microchem. J. 158, 105222 (2020). DOI: 10.1016/j.microc.2020.105222
(7) D. Pavel, R.R. Miguel, V. Vlastimil, and R.J. António, Talanta 217, 121068 (2020). DOI: 10.1016/j.talanta.2020.121068
(8) K.M. Lea, B. Maja, and R. Njegomir, Acta Pharmaceutica 71(4), 619–630 (2021).
(9) N.G. Yasri, H. Seddik, and M.A. Mosallb, Arab. J. Chem. 8(4), 487–494 (2015).
(10) T.T. Ma, D.Z. Wang, Y.H. Zhang, S. Zhang, S.H. Sun, X. Li, and J.X. Zhang, Anal. Lett. 54(3), 331–346 (2021).
(11) C.J. Keun, J. Soc. Cosmetic. Scientists. Korea. 42(1), 65–73 (2016).
(12) F.B.de.F. Rezende, A.M.de.S.S. Cheibub, A.D.P. Netto, and F.F.de.C. Marques, Microchem. J. 134, 383–389 (2017).
(13) M.Y. Wang, Z.B. Guo, H.Y. Guan, and M. Li, Key. Eng. Mater. 4348, 355–359 (2017).
(14) F.A. Lobo, T.M.O. Santos, K.M. Vieira, V.M. Osório, and J.G. Taylor, Drug. Test. Anal. 7(9), 848–852 (2015).
(15) M. Hladová, J. Martinka, and P. Rantuch, Res. Pap. Fac. Mater. Sci. Technol. Slovak. U. Technol. 27(44), 105–120 (2019).
(16) R. Giesen, T. Schripp, D. Markewitz, B. Meyer, H. Schwab, E. Uhde, and T. Salthammer, Anal. Lett. 49(10), 1613–1621 (2016).
(17) T. Zhang, Q.Q. Xu, L.Y. Chen, and Y.F. Zheng, Guangzhou Chem. Ind. (In Chinese) 41(12), 163–164 (2013).
(18) X.Z. Shen and Z.C. Liu, Metall. Anal. (In Chinese) 2008(10), 53–56 (2013).
(19) Z.P. Li, X.R. Duan, and C.H. Liu, Anal. Biochem. 351(5), 18–25 (2006).
(20) X.K. Wang, J.G. Wang, P.Q. Guo, W.L. Guo and G.L. Li, Ultrason. Sonochem. 15(4), 357–363 (2008).
(21) T.C. An, X.H. Zhu, and Y. Xiong, Chemosphere 46(6), 897–903 (2002).
Wang Zhao-qun, Sheng Li, Han Feng, Zhang Xin, and Su Bi-quan is with the College of Chemistry and Chemical Engineering at Lanzhou Jiaotong University, in Lanzhou, China. Li Hai-su is with the Key Institute of Lightwave Technology at Beijing Jiaotong University, in Beijing, China. Direct correspondence to Wang Zhao-qun at zq960805@sina.com. ●

New Study Reveals Insights into Phenol’s Behavior in Ice
April 16th 2025A new study published in Spectrochimica Acta Part A by Dominik Heger and colleagues at Masaryk University reveals that phenol's photophysical properties change significantly when frozen, potentially enabling its breakdown by sunlight in icy environments.
Tracking Molecular Transport in Chromatographic Particles with Single-Molecule Fluorescence Imaging
May 18th 2012An interview with Justin Cooper, winner of a 2011 FACSS Innovation Award. Part of a new podcast series presented in collaboration with the Federation of Analytical Chemistry and Spectroscopy Societies (FACSS), in connection with SciX 2012 ? the Great Scientific Exchange, the North American conference (39th Annual) of FACSS.
New Fluorescence Model Enhances Aflatoxin Detection in Vegetable Oils
March 12th 2025A research team from Nanjing University of Finance and Economics has developed a new analytical model using fluorescence spectroscopy and neural networks to improve the detection of aflatoxin B1 (AFB1) in vegetable oils. The model effectively restores AFB1’s intrinsic fluorescence by accounting for absorption and scattering interferences from oil matrices, enhancing the accuracy and efficiency for food safety testing.
Can Fluorescence Spectroscopy Evaluate Soil Dissolved Organic Matter Dynamics?
February 20th 2025A new study published in Chemical Engineering Journal by researchers from Northeast Agricultural University in China reveals that biochar aging, influenced by environmental factors like UV exposure and wet-dry cycles, alters dissolved organic matter composition and affects its effectiveness in remediating cadmium-contaminated soil.
Geographical Traceability of Millet by Mid-Infrared Spectroscopy and Feature Extraction
February 13th 2025The study developed an effective mid-infrared spectroscopic identification model, combining principal component analysis (PCA) and support vector machine (SVM), to accurately determine the geographical origin of five types of millet with a recognition accuracy of up to 99.2% for the training set and 98.3% for the prediction set.