The Benzene Fingers, Part I: Overtone and Combination Bands
Spectroscopy
This installment begins with a needed discussion on the theory behind the three different types of infrared bands, how to recognize them, and how to use them to help you interpret spectra. Continuing on from the last column, this knowledge is used to help better distinguish mono- and di-substituted benzene rings from each other.
This installment begins with a much needed discussion on the theory behind the three different types of infrared bands, how to recognize them, and how to use them to help you interpret spectra. Continuing on from the last column, this knowledge is used to help better distinguish mono- and disubstituted benzene rings from each other.
In the last installment (1), we discussed how to distinguish mono- and disubstituted benzene rings from each other. We used the position of the aryl C-H out-of-plane bending vibration (“C-H wag”) between 1000 and 700 cm-1, and the presence or absence of the intense ring bending peak at 690 ± 10 cm-1. This information is summarized in Table I.
What I didn’t discuss in the last column is that there is a problem with Table I. If for a given molecule the C-H wag falls between 770 and 750 cm-1 and there is a ring bend present, the molecule could be mono- or meta-substituted. This means we cannot distinguish mono- and meta-substituted molecules from each other in this circumstance. What are we to do? Are there any other bands in the spectra of substituted benzene rings that can help us?
In the spectra of substituted benzene rings there exists a series of peaks between 2000 and 1650 cm-1 whose number and patterns correlate with a substitution pattern. These peaks are formally called the summation bands (2), but I like to call them the benzene fingers, since they form a pattern reminiscent of the fingers on a human hand. An example of the benzene fingers are labeled in Figure 1.
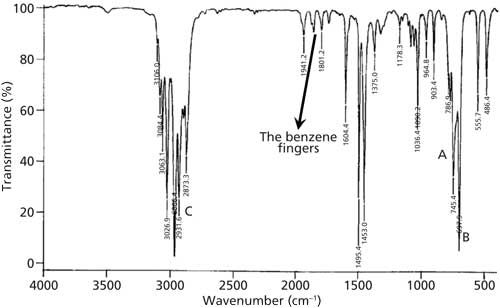
Figure 1: The IR spectrum of a substituted benzene ring with the benzene fingers labeled.
The benzene fingers are a series of overtone and combination bands, which at this point will not mean much to you since we have not defined these terms. That is the purpose of the next section.
Fundamental, Overtone, and Combination Bands
There are essentially three different types of infrared (IR) peaks: fundamental, overtone, and combination bands. To obtain an understanding of these peak types, we need to have a hasty brush with quantum mechanics.
Recall from the first installment of this column (3) that when a molecule absorbs an IR photon it makes a spectroscopic transition from a lower to a higher vibrational energy level. This transition occurs because the vibrational energy levels in a molecule are quantized. This means that molecules can’t have any random amount of vibrational energy, but only specific allowed vibrational energies. Quantized energy levels are features of bound microscopic systems such as atoms and molecules (4). Figure 2 shows a hypothetical vibrational energy level diagram for the O-H stretching vibration of water.
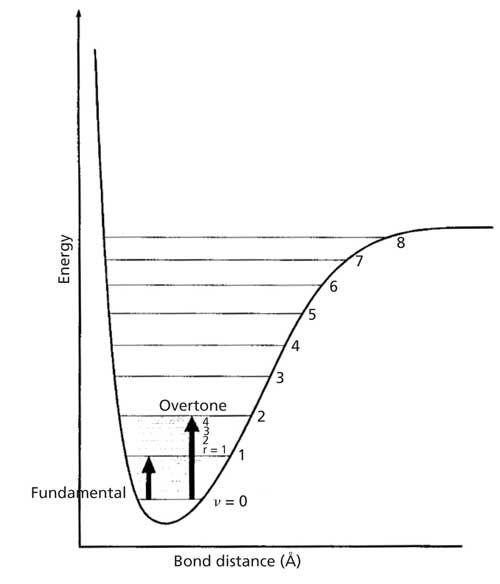
Figure 2: A hypothetical energy level diagram for the O-H stretch of water.
The x-axis of the diagram is O-H bond length in angstroms, and the y-axis is vibrational energy measured in wavenumbers. The lines represent the allowed O-H stretching vibrational energy levels. The numbers accompanying each vibrational energy level are called vibrational quantum numbers, and are denoted with a v. Note that the energy levels are numbered 0, 1, 2, and so on. These energy levels are approximately equally spaced and are about 3500 cm-1 apart. Thus, water is only allowed to have O-H vibrational energies of ~3500 cm-1, ~7000 cm-1, and so forth. An energy level diagram like Figure 2 can be drawn for most molecular vibrations.
Recall (3) that the peak positions in an IR spectrum are given by equation 1:
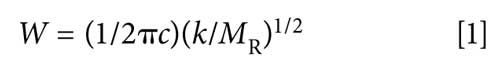
where W is the peak wavenumber position in cm-1, c is the speed of light, k is the force constant, and MR is the reduced mass. This equation, in my opinion, is the most important one in IR spectroscopy. It quantitates the correlation between peak position and molecular structure, it is the reason why two molecules have different chemical structures if they have different IR spectra, and it is why we can use IR spectra as a chemical fingerprint.
Molecular vibrational energy level spacings like those in Figure 2 are determined by equation 1. For example, there is an O-H stretching peak at ~3500 cm-1 in the spectrum of water from it undergoing a spectroscopic transition from the v = 0 to v = 1 level. The reason its energy levels are ~3500 cm-1 are because of the reduced masses and force constants of the molecule determined that spacing per equation 1. For C-H stretches the force constants and reduced masses give energy levels that are about 3000 cm-1 apart. For methyl group umbrella modes the force constants and reduced masses give energy levels that are approximately 1375 cm-1 apart.
Whenever a molecule undergoes a vibrational spectroscopic transition between the v = 0 and v = 1 levels the resulting transition is called a fundamental transition, and the resultant IR peak is called a fundamental peak. A fundamental transition is illustrated by the smaller dark arrow in Figure 2. Fundamental bands are inherently intense, make up most of the peaks seen in mid-IR spectra, and comprise most of the diagnostically useful peaks we have been and will be studying for interpretation purposes.
An overtone band is the result of any spectroscopic transition from v = 0 to v = 2,3,4 . . . or higher. An example of an overtone transition is shown in Figure 2. The “first overtone” involves a v = 0 to v = 2 transition, and falls at about twice the wavenumber of the fundamental. Figure 3 shows the IR spectrum of methyl ethyl ketone with examples of a fundamental and overtone band labeled. Note that the overtone is about 10 times less intense than the fundamental. In general, overtone bands are 10–100 times less intense than fundamental bands (4). Thus, only overtones of very intense fundamentals will typically appear in IR spectra. For fundamentals above 2000 cm-1 their overtones will mostly appear above 4000 cm-1 in the near-infrared (NIR) part of the electromagnetic spectrum. Since overtones are weak they are generally not diagnostically useful IR features.
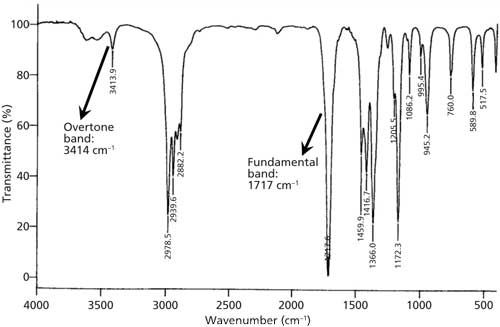
Figure 3: The IR spectrum of methyl ethyl ketone, with examples of fundamental and overtone bands marked.
CLICK QUIZ ANSWER TO ENLARGE. IF QUIZ IS STILL TOO SMALL, CLICK ONCE MORE AND IT WILL ENLARGE FURTHER.
In addition to overtones, when a molecule absorbs IR light more than one vibration can be excited at the same time. These types of IR features are called combination bands because they involve the excitation of a combination of vibrations. The infrared spectrum of liquid water, seen in Figure 4, shows examples of how two fundamental bands can combine to give a combination band.
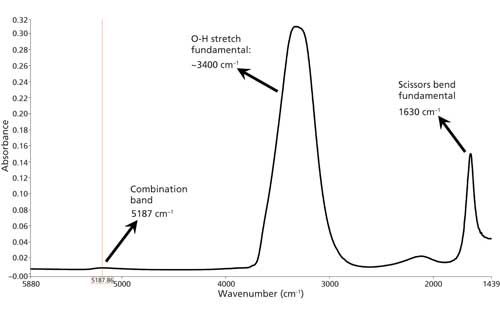
Figure 4: The IR spectrum of liquid water with the scissors vibration, O-H stretch, and combination band marked.
The water spectrum exhibits a scissors bending fundamental at 1630 cm-1, and an O-H stretch fundamental at ~3400 cm-1. The combination band, because of the excitation of both these vibrations at the same time, is at 5187 cm-1. Note how small the combination band is compared to the fundamentals. Also note that this combination band is in the NIR. Many NIR features are overtone or combination bands. Because they are not intense, combination bands are generally not diagnostically useful.
So, if both overtone and combination bands are not diagnostically useful why did I spend so much time introducing them to you? Because the benzene fingers are a rare breed, a series of diagnostically useful overtone and combination bands that will allow us to determine benzene ring substitution patterns when Table I fails us. We will complete our discussion of the benzene fingers in the next installment.
References
- B.C. Smith, Spectroscopy31(3), 34–37 (2016).
- B.C. Smith, Infrared Spectral Interpretation: A Systematic Approach (CRC Press, Boca Raton, Florida, 1999).
- B.C. Smith, Spectroscopy30(1), 16–23 (2015).
- B.C. Smith, Quantitative Spectroscopy: Theory and Practice (Academic Press, Boston, Massachusetts, 2002).
- B.C. Smith, Spectroscopy 31(5), 36–39 (2016).
- B.C. Smith, Spectroscopy30(7), 26–31, 48 (2015).

Brian C. Smith, PhD, is a Senior Infrared Product Specialist for PerkinElmer, based in San Jose, California. Before joining PerkinElmer, he ran his own FT-IR training and consulting business for more than 20 years, and taught thousands of people around the world how to improve their FT-IR analyses and interpret infrared spectra. Dr. Smith has written three books on infrared spectroscopy: Fundamentals of FTIR and Infrared Spectral Interpretation, both published by CRC Press, and Quantitative Spectroscopy: Theory and Practice published by Academic Press. He has published a number of papers in peer-reviewed journals and is a co-inventor on a patent for an FT-IR method to monitor dust exposure in coal mines.

AI Shakes Up Spectroscopy as New Tools Reveal the Secret Life of Molecules
April 14th 2025A leading-edge review led by researchers at Oak Ridge National Laboratory and MIT explores how artificial intelligence is revolutionizing the study of molecular vibrations and phonon dynamics. From infrared and Raman spectroscopy to neutron and X-ray scattering, AI is transforming how scientists interpret vibrational spectra and predict material behaviors.
Real-Time Battery Health Tracking Using Fiber-Optic Sensors
April 9th 2025A new study by researchers from Palo Alto Research Center (PARC, a Xerox Company) and LG Chem Power presents a novel method for real-time battery monitoring using embedded fiber-optic sensors. This approach enhances state-of-charge (SOC) and state-of-health (SOH) estimations, potentially improving the efficiency and lifespan of lithium-ion batteries in electric vehicles (xEVs).
New Study Provides Insights into Chiral Smectic Phases
March 31st 2025Researchers from the Institute of Nuclear Physics Polish Academy of Sciences have unveiled new insights into the molecular arrangement of the 7HH6 compound’s smectic phases using X-ray diffraction (XRD) and infrared (IR) spectroscopy.