A Corrosive Liquid Investigated by Diamond ATR Infrared Spectroscopy

Experimental
Infrared spectra were collected on an FT-IR spectrometer equipped with the Harrick DiaMaxATR™ single-reflection high-throughput diamond ATR accessory (see Figure 1). The system was purged to remove water vapor and CO2. Spectra were collected at 4 cm-1 resolution and the signal was averaged over 16 scans. The spectra were referenced to the clean ATR crystal.
Figure 1: The DiaMaxATR Diamond ATR.

The sample used here was Easy-Off™ Heavy Duty Oven Cleaner (Reckitt Benckiser). Two drops of the oven cleaner were placed on the center of the ATR crystal and spectra were collected at 20 scans per s over a period of 60 min.
Results and Discussion
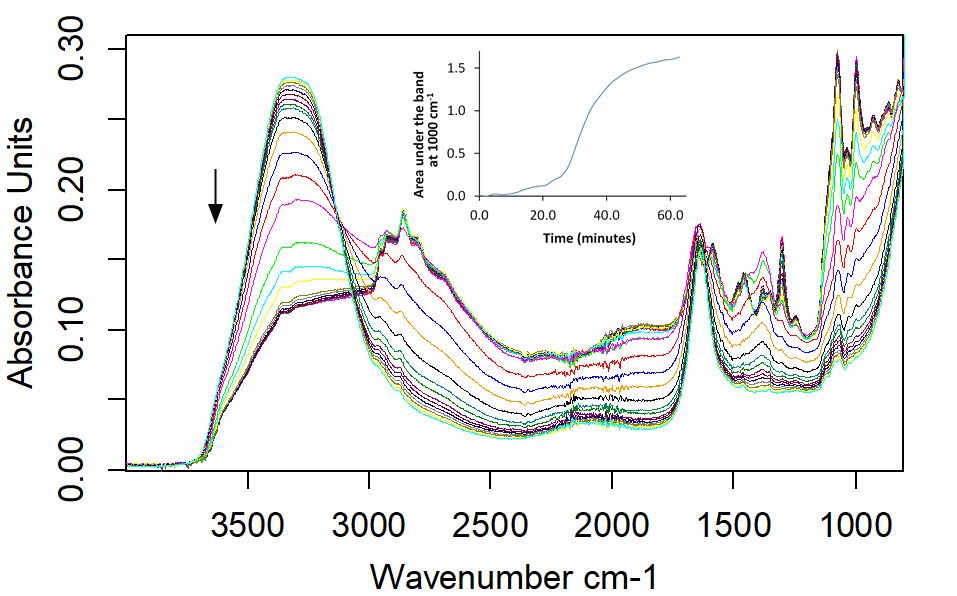
Figure 2 shows the series of FT-IR spectra acquired over the period of 60 min, showing water evaporating from the oven cleaner and concentrating the remaining ingredients. The MSDS for the oven cleaner indicates that it is composed of 2-(2-Butoxyethoxy)ethanol, 2-amino-ethanol, sodium hydroxide, and liquefied petroleum gases. As it dries, the OH stretching and bending vibrations around 3300 and 1645 cm-1 respectively decrease, showing the evaporation of water present with sodium hydroxide. The bands at 2958, 2930, 2862, and 2800 cm-1, due to CH stretches, increase as the oven cleaner becomes more concentrated, as do the amine bands at 1658 and 1590 cm-1. Presumably the volatile petroleum components are also evaporating. But the decrease in the volatile organics is more difficult to discern, due to the increased concentration of the nonvolatile compounds. Note that the rate at which the oven cleaner concentrates could be measured by looking at peak areas, as demonstrated by the inset graph for the C=C transition at 1000 cm-1
Figure 2: ATR spectra of oven cleaner over time. The arrow indicates increasing time, and the inset shows the change in concentration of C=C species with time.
Conclusion
Because diamond is a more chemically durable material, it can be effectively used as an ATR crystal for some corrosive samples. This has been demonstrated here using the Harrick DiaMaxATR to measure the ATR spectra of oven cleaner, as it becomes increasingly concentrated due to evaporation of the solvent therein.
Harrick Scientific Products, Inc.
141 Tompkins Ave., 2nd Floor, Pleasantville, NY 10570
tel. (800) 248-3847
Website: www.harricksci.com

New Study Reveals Insights into Phenol’s Behavior in Ice
April 16th 2025A new study published in Spectrochimica Acta Part A by Dominik Heger and colleagues at Masaryk University reveals that phenol's photophysical properties change significantly when frozen, potentially enabling its breakdown by sunlight in icy environments.
Advanced Raman Spectroscopy Method Boosts Precision in Drug Component Detection
April 7th 2025Researchers in China have developed a rapid, non-destructive Raman spectroscopy method that accurately detects active components in complex drug formulations by combining advanced algorithms to eliminate noise and fluorescence interference.